From fossils to conservation: an overview of Antarctic and sub-Antarctic penguins
Raven Quilestino-Olario
Penguins are among the most distinctive birds of the Antarctic and sub-Antarctic, and their story spans from extinct “giant” fossil species to the penguins that breed today. This overview brings together evidence from fossils, ancient colony remains, chemical records, and modern monitoring to place today's population changes in a long environmental context. Fossils from Seymour Island and other Southern Hemisphere sites show that penguins have persisted for more than 60 million years, including during much warmer periods, followed by major changes as Antarctica cooled and became dominated by ice. Records from past colonies and genetic studies show that penguin populations repeatedly expanded, contracted, and shifted their ranges as sea ice and ocean conditions changed over thousands of years, highlighting how strongly penguins depend on sea ice and marine food webs. Eight Antarctic and sub-Antarctic species are assessed, summarizing current status and pressures using recent conservation assessments alongside ecological and satellite-based studies. Trends differ strongly by region: some populations, such as gentoo penguins in parts of the Antarctic Peninsula, have expanded or shifted their range, while others have declined or appear highly sensitive to changes in food availability. Looking ahead, risk is shaped by connected processes rather than warming alone. Ice-dependent species are most vulnerable where reliable sea ice is disappearing or breaking up earlier, whereas krill-dependent species face added risk where climate-driven changes in krill overlap with concentrated fishing. New tools that are improving monitoring and forecasting are highlighted, including satellite and aerial colony surveys, animal tracking, remote cameras, and improved krill mapping, alongside key remaining uncertainties such as juvenile survival, winter behavior, and the fine-scale conditions near colonies. Overall, this synthesis emphasizes a central contrast: penguins have shown resilience over deep time, yet their near-future persistence may be constrained by the speed and combined pressures of human-driven change.
- Article
(4892 KB) - Full-text XML
- BibTeX
- EndNote
Penguins are among the most recognizable animals of the Antarctic and sub-Antarctic region. Beyond their appeal as symbols of the southern oceans, they play important roles in marine food webs as mid-level predators that link plankton and krill to higher predators such as seals and whales (Croxall and Prince, 1980; Fraser and Trivelpiece, 1996). Their life cycles are closely tied to sea ice, ocean fronts, and prey availability, which makes them sensitive to environmental change and human activities in the Southern Ocean (Ainley, 2002; Trathan et al., 2020). Because their populations respond measurably to shifts in sea ice conditions and marine productivity, penguins are widely used as indicators of ecosystem change in Antarctic and sub-Antarctic environments (Boersma, 2008; Forcada and Trathan, 2009).
Penguin conservation has therefore received sustained attention. As flightless seabirds that breed on land or sea ice but forage at sea, they are exposed to multiple pressures that affect breeding habitat, food supply, and reproductive timing (Williams, 1995; Lynch et al., 2012). Several species have already shown clear responses to recent environmental change. Emperor penguins have experienced breeding failures following early sea ice breakup (Fretwell and Trathan, 2009; Trathan et al., 2020), while gentoo penguins have expanded southward into parts of the Antarctic Peninsula formerly dominated by Adélie penguins (Ducklow et al., 2007; Hinke et al., 2007). In contrast, some krill-dependent sub-Antarctic species, including macaroni and southern rockhopper penguins, have declined in several regions (Croxall et al., 2002; Trathan et al., 2006).
Global assessments by the International Union for Conservation of Nature (IUCN) and BirdLife International provide the most recent evaluations of the conservation status of penguins (BirdLife International, 2023; IUCN, 2023). At the same time, paleontological discoveries, geochemical analyses, and ecological modeling continue to refine our understanding of the origins of penguins, their environmental history, and their projected responses to change (Ksepka and Clarke, 2010; Trathan et al., 2020). These different lines of evidence are usually treated separately, spanning fields from paleobiology to conservation ecology.
This overview brings these perspectives together for Antarctic and sub-Antarctic penguins. By linking fossil history, modern distribution and status, and future conservation challenges, it places present-day population trends in a long-term environmental context. Viewed across this time span, penguins illustrate both evolutionary resilience and increasing sensitivity to rapid, human-driven change. The aim is to provide a clear and accessible overview of how knowledge from fossils to conservation informs our understanding of these key Southern Ocean species.
This article is based entirely on published and openly available literature. The core sources are the most recent global conservation assessments from the IUCN and BirdLife International, which provide standardized information on penguin population size, status, and threats (BirdLife International, 2023; IUCN, 2023). These are complemented by peer-reviewed studies published from the early 20th century to the present.
Rather than generating new datasets, this overview brings together results from paleontology, geochemistry, ecology, and conservation science to interpret modern penguin status and trends in a longer environmental and evolutionary context. This overview synthesizes fossil records, geochemical proxies, demographic studies, and recent monitoring and modeling to highlight recurring patterns, current pressures, and the main uncertainties that still limit prediction across Antarctic and sub-Antarctic species.
Modern trends are easier to interpret in a longer context. The fossil record and colony archives reveal when key penguin traits emerged and how populations shifted as Antarctica cooled and sea ice regimes changed. This context helps to frame present-day observations in terms of both resilience and sensitivity.
3.1 Fossil record and evolutionary history
Penguins have a long evolutionary history that reaches back more than 60 million years to the early Paleocene. The oldest widely accepted stem (ancestral) penguins, Waimanu manneringi and W. tuatahi (later reassigned to Muriwaimanu tuatahi), come from the Waipara Greensand Formation in the Canterbury region of New Zealand (Slack et al., 2006; Mayr et al., 2020, 2025). These early fossils already show adaptations for wing-propelled diving, suggesting that the shift from flight to underwater “flight” occurred early in penguin evolution.
Fossil discoveries on Seymour Island, near the Antarctic Peninsula, are especially important because they document penguins living along Antarctic coasts during the warmer climates of the Eocene and Oligocene. They include large extinct species such as Palaeeudyptes gunnari and Anthropornis nordenskjoeldi, dated to about 34–37 million years ago (Marples, 1953; Jadwiszczak, 2006). These “giant penguins”, in some cases approaching 2 m in height, show that penguins were already diverse and successful in Antarctica before the modern ice sheet system became fully established (Martin, 2009; Stilwell and Long, 2011).
Recent work continues to refine this record. A newly described Eocene specimen from Seymour Island adds further evidence for high morphological diversity among early Antarctic penguins and also highlights ongoing uncertainty in how some large-bodied taxa are separated when size is used as a primary criterion (Xia et al., 2024). Later fossils from South America and New Zealand during the Miocene and Pliocene indicate dispersal and diversification beyond Antarctica (Ksepka and Clarke, 2010). By the Pliocene, penguins had expanded into colder, ice-influenced marine environments more comparable to those they occupy today (Martin, 2009; Stilwell and Long, 2011). A recent synthesis of penguin biogeography supports a New Zealand origin followed by expansion into Antarctica in the Eocene, a major turnover during Antarctic cooling, and later re-expansions of modern lineages across the Southern Hemisphere (Pelegrín and Acosta Hospitaleche, 2022). Together, the fossil record shows both the early first occurrences of penguins and their capacity to persist across major shifts in climate and ocean conditions, providing long-term context for modern conservation concerns (see Fig. 1).
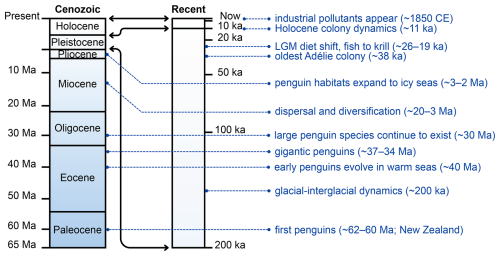
Figure 1Geologic timeline of penguin evolution and environmental archives from the Paleocene to the present. The vertical axis represents time, with bands highlighting the Paleocene to the modern periods. Key milestones include the earliest stem penguins in the Paleocene (Waimanu manneringi and W. tuatahi (= Muriwaimanu tuatahi) from the Waipara Greensand Formation, Canterbury, New Zealand), giant fossil species on Seymour Island during the Eocene–Oligocene (Marples, 1953; Jadwiszczak, 2006; Xia et al., 2024) when Antarctic seas were warmer and largely ice free (Martin, 2009; Stilwell and Long, 2011), and diversification and dispersal during the Miocene–Pliocene (Ksepka and Clarke, 2010). Late Quaternary archives include radiocarbon-dated colony remains (bones, eggshells, guano) that record occupation history and environmental change, such as the oldest known Adélie colony at Cape Adare (∼ 38 ka) (Baroni and Orombelli, 1994; Emslie, 2001), diet shifts during the Last Glacial Maximum (∼ 26–19 ka) inferred from stable isotopes (Emslie and Patterson, 2007; Sun et al., 2000), and guano and eggshell records tracing Holocene colony dynamics (Huang et al., 2009). Industrial-era pollutants (since the determination of colonies after ∼ 1850 CE; Xu et al., 2020) illustrate the most recent environmental signals preserved in penguin tissues and deposits. Abbreviations: Ma = million years ago; ka = thousand years ago; CE = Common Era; LGM = Last Glacial Maximum.
3.2 Pleistocene records
Evidence from the Pleistocene (2.6 Ma to 11.7 ka) shows that penguin populations repeatedly expanded and contracted as ice sheets advanced and retreated. Much of this evidence comes from subfossil remains preserved in raised beaches and abandoned colonies, together with genetic data that record past population bottlenecks and re-expansions. Ancient DNA extracted from penguin remains has been used to compare past and modern genetic diversity and to test whether regional colonies persisted locally or were repeatedly re-founded after glacial maxima (Ritchie et al., 2004; Emslie, 2021). For example, material from the Scotia Arc region (including the South Shetlands and South Orkneys) indicates that genetic structure and connectivity shifted across glacial–interglacial cycles, consistent with changing habitat availability and dispersal corridors (Ritchie et al., 2004).
Sedimentary and geochemical records provide complementary evidence for postglacial recolonization. In East Antarctica, an ornithogenic sediment core from the Vestfold Hills suggests that Adélie penguins began colonizing newly ice-free ground shortly after local deglaciation, with occupation beginning around 14.6 ka and increasing through the Holocene (Gao et al., 2018). This supports the broader view that the emergence of accessible breeding habitat is a first-order control on colony establishment following glacial retreat.
Genomic studies reinforce this refugia and recolonization picture at a wider taxonomic scale. Genome-wide demographic reconstructions across living penguin species frequently indicate population declines around the Last Glacial Maximum (ca. 26–19 ka), consistent with reduced marine productivity and fragmented breeding habitat, followed by postglacial recovery in many lineages (Vianna et al., 2020; Cole et al., 2022). For emperor penguins specifically, phylogeographic analyses suggest that suitable breeding habitat during the Last Glacial Maximum was fragmented, with populations persisting in refugia before expanding again as sea ice conditions relaxed during the Holocene (Younger et al., 2015). Together, the Pleistocene record shows that climate-driven habitat change leaves measurable signatures in penguin population size, distribution, and genetic diversity, providing a long-term context for interpreting modern change (Fig. 1).
3.3 Holocene colonies
The Holocene (last 11 700 years) provides a particularly rich archive of penguin remains. Cape Adare in the Ross Sea hosts the oldest known Adélie penguin colony, with remains dated to around 38 000 years ago and continuous evidence of occupation through the Holocene (Baroni and Orombelli, 1994; Emslie and Patterson, 2007). Some Antarctic colony sites have therefore persisted for millennia, indicating long-term stability where suitable nesting habitat and access to open water were maintained.
Radiocarbon-dated eggshells, bones, and guano deposits from the Ross Sea and Antarctic Peninsula record colony establishment, abandonment, and shifts in distribution in response to regional climate and sea ice variability (Emslie, 2001; Huang et al., 2009). Stratified Holocene deposits from the northern Antarctic Peninsula show that Adélie, gentoo, and chinstrap penguins all bred in the region during early Holocene warming and deglaciation, followed by local contraction during later cooling phases (Emslie et al., 2020).
Colony histories can also reveal large-scale demographic change. At Cape Adare, radiocarbon data indicate the rise of a mid-Holocene Adélie “supercolony” exceeding 500 000 breeding pairs, followed by contraction linked to changing sea ice and coastal conditions (Emslie et al., 2018). New genomic approaches further expand these archives. Sedimentary DNA from Ross Sea colony soils spanning the last 6000 years records shifts in the penguin diet, population diversity, and even faunal turnover events, demonstrating how colony sediments preserve ecological responses to climate and sea ice variability (Wood et al., 2025).
In several coastal regions, penguin and seal bones have been dated together with other coastal deposit materials to constrain the timing of raised beaches and glacier advances during the late Holocene (Clapperton and Sugden, 1988). More recent work shows that warming-driven snowmelt can expose previously frozen penguin remains, revealing earlier occupation phases and past periods of higher marine productivity during the mid-Holocene (Emslie, 2021). Together, these records demonstrate that penguin remains contribute to both ecological reconstructions (colony persistence, relocation, diet, and population change) and geomorphic reconstructions (dated coastal deposits and glacier fluctuations), processes summarized in Fig. 1.
3.4 Diet, trophic dynamics, and multi-proxy archives
Penguin remains are valuable natural archives of past ecosystems and environments because they preserve information on diet, food web structure, and environmental conditions at colony sites. Stable isotope measurements of carbon (δ13C) and nitrogen (δ15N) in eggshells, bone collagen, feathers, and guano show that penguin diets and trophic dynamics can shift across major climate transitions. For example, during the Last Glacial Maximum (ca. 26–19 ka), Adélie penguins relied more heavily on fish, whereas Holocene diets shifted toward krill, consistent with changes in sea ice conditions and marine productivity (Sun et al., 2000; Emslie and Patterson, 2007). Importantly, recent work using compound-specific (amino-acid) δ15N from Holocene eggshell archives shows that part of the long-term decline in bulk δ15N values can reflect changes in baseline biogeochemical cycling at the base of the food web, not only changes in trophic position (Michelson et al., 2023). Multi-proxy records from ornithogenic soils combine hard prey remains with stable isotopes and can reveal shifts in prey use through time at colony sites (Kalvakaalva et al., 2020). These dietary signals also respond to modern sea ice variability. Isotopic evidence for prey switching between euphausiids and pelagic fishes in penguins coincides with interannual sea ice differences, and has broader ecosystem implications because penguins and seals can transport limiting nutrients to coastal environments (Wing et al., 2021). Beyond Antarctica, late-Holocene isotope records also show that ecosystem restructuring and historic exploitation of marine megafauna can alter the penguin trophic position after accounting for baseline change, including in Magellanic penguins (Bas et al., 2023).
In addition to isotopes, penguin-derived materials support chronologies and environmental reconstructions. Penguin bones are widely used for radiocarbon dating, often together with driftwood, seaweed, and shells, to date beaches, moraines, and glacier fluctuations (Clapperton and Sugden, 1988; Emslie, 2001). A recent example from Cape Irizar in the Ross Sea shows how snowmelt exposed well-preserved Adélie penguin bones and carcasses from colonies occupied between ca. 5000 and 800 cal. yr BP (Emslie, 2021). Radiocarbon ages and elevated δ13C values in bone collagen indicate a period of enhanced marine productivity during a mid-Holocene occupation optimum, and the exposure of these remains also illustrates how recent warming can re-reveal long-frozen biological archives. Guano deposits can record colony presence and food web structure through organic matter and geochemistry, and have been used to reconstruct dietary shifts (Huang et al., 2009) and detect heavy metals such as mercury and lead that reflect both natural processes and, in recent centuries, human pollution (Baroni and Orombelli, 1994; Sun et al., 2000). Recent syntheses highlight vertebrate colonies, including penguin rookeries, as key Antarctic biological archives that complement ice cores and marine sediments by recording ecological responses to climate variability through time (Strugnell et al., 2022). Together, these archives show how penguin-derived records can be used to reconstruct environmental change across both terrestrial ice-free areas and adjacent coastal marine systems.
3.5 Modern perspectives
Over the last few centuries, and especially since the industrial era, penguin colonies have continued to record environmental change at the land–sea boundary. Feathers, eggshells, and guano layers can preserve contaminants such as lead and mercury, as well as organic and geochemical signals that reflect shifts in marine productivity and food web structure (Baroni and Orombelli, 1994; Sun et al., 2000). In addition, repeated sampling of the same colony landscapes allows older, long-term records to be interpreted alongside present-day observations, linking archive evidence to the conditions penguins experience today.
In this context, penguins can act as sentinels of ecological change in a specific sense. They integrate changes in sea ice, prey fields, and ocean productivity through measurable outcomes such as breeding success, diet, and colony occupancy, and they also leave physical and chemical traces of those changes in their remains and colony sediments. This makes penguin archives a bridge between paleoenvironmental reconstruction and conservation science, because the same systems that shaped colonies in the past are the ones now being rapidly altered by human-driven warming and direct impacts (Boersma, 2008; Forcada and Trathan, 2009).
3.6 Summary
The evidence reviewed in this section shows that penguins provide a rare, time-continuous archive of Southern Ocean change. Fossils document early origins and major radiations, including diverse large-bodied forms during warm Eocene conditions, followed by turnover and persistence as Antarctica cooled and became ice dominated. Quaternary records and genetic evidence show repeated range shifts and demographic change through glacial cycles, while Holocene colony deposits and geochemistry record fluctuations in colony occupancy, diet, and ecosystem structure as sea ice and productivity varied. In the modern era, penguin-derived materials also preserve signals of direct human influence, including industrial contaminants.
Together, these archives support two linked conclusions that motivate the rest of the paper. First, penguins have repeatedly persisted through large natural environmental shifts over millions of years, which demonstrates long-term resilience and ecological flexibility. Second, the same records show that penguin populations are tightly coupled to sea ice, prey availability, and oceanographic change, which makes them vulnerable when these drivers shift rapidly and simultaneously. This contrast between deep-time persistence and sensitivity to fast, human-driven change provides the long-term context for interpreting present-day trends and conservation risk.
The narrative then moves to the present. Today's penguins are the extant outcome of past diversification and turnover, and they occupy a wide range of Southern Ocean habitats. Here, eight Antarctic and sub-Antarctic breeders are used as focal species to compare status, pressures, and the mechanisms most likely to shape near-future change.
4.1 The penguins
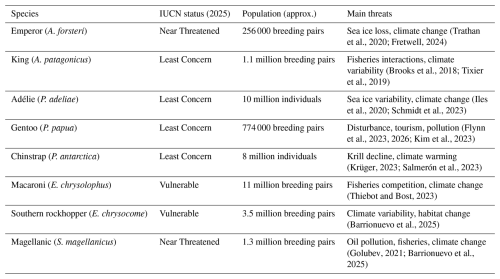
This synthesis focuses on eight penguin species that breed in the Antarctic and sub-Antarctic region (Fig. 2): emperor (Aptenodytes forsteri), king (A. patagonicus), Adélie (Pygoscelis adeliae), gentoo (P. papua), chinstrap (P. antarctica), macaroni (Eudyptes chrysolophus), southern rockhopper (E. chrysocome), and Magellanic (Spheniscus magellanicus). Together they span sea ice breeders, continental coastal breeders, and sub-Antarctic island species linked to ocean fronts and shelf systems (Williams, 1995; Ainley, 2002; Kristan, 2021; De Roy et al., 2022). Throughout this section, “population size” refers to global estimates reported by the IUCN and BirdLife, but these values are compiled from mixed methods and do not have equal precision across species and regions. Likewise, “increasing” or “declining” is used to describe the dominant direction reported in recent assessments or regional long-term datasets, not a uniform trend across the entire range. This distinction matters because many species show opposite trajectories in different sectors, reflecting local sea ice conditions, prey availability, and human activity. Penguins are therefore useful indicators of Southern Ocean change because they translate environmental variability into measurable outcomes such as breeding success, colony occupancy, diet, and distribution shifts.

Figure 2Representative Antarctic and sub-Antarctic penguin species included in this overview: (a) emperor (A. forsteri), (b) king (A. patagonicus), (c) Adélie (P. adeliae), (d) gentoo (P. papua), (e) chinstrap (P. antarctica), (f) macaroni (E. chrysolophus), (g) southern rockhopper (E. chrysocome), and (h) Magellanic (S. magellanicus). Approximate adult maximum height (m) and body mass (kg) are compiled from species summaries by the World Wide Fund for Nature (WWF, 2026). Penguin images are publication-licensed stock illustrations from Canva (Canva for Education), assembled by the author; used under the Canva Content License Agreement (https://www.canva.com/en_in/policies/content-license-agreement/, last access: 16 March 2026) for online or electronic publication and for non-commercial purposes.
4.1.1 Emperor penguins (Antarctic)
Emperor penguins (Fig. 2a) breed on seasonal fast ice and are the only penguin species that raises chicks through the Antarctic winter. This life history ties breeding success to sea ice timing and stability. Sea ice affects emperors directly by providing the breeding platform and indirectly by shaping access to prey through polynyas, ice edges, and the seasonal distribution of fish and krill.
Recent observations show how quickly early sea ice loss can translate into breeding failure. In 2022, record-low sea ice in the Bellingshausen Sea was linked to widespread colony failure when breakup occurred before the fledging period (Fretwell et al., 2023). Similar chick loss has also been documented after an unusually early breakup of fast ice at a high-latitude colony, indicating that even very southerly sites can be exposed to damaging breakup events (Schmidt and Ballard, 2020). Over longer periods, satellite-based assessments suggest that declines in some regions may already be exceeding earlier model expectations (Fretwell et al., 2025), and remote-sensing time series indicate an overall downward tendency in global indices over the last decade, although uncertainty remains because colony occupancy and detectability vary within and among seasons (Labrousse et al., 2022; LaRue et al., 2024). New phenology-based methods can help to reduce this uncertainty by estimating breeding pairs and fledging success from sparse imagery, which strengthens monitoring in remote areas where ground counts are rare (Winterl et al., 2024). Recent studies also show that emperor breeding habitat can shift and fragment under climate extremes (Lin et al., 2025) and that early-life survival is strongly tied to body condition and environmental access to food during and after fledging (Le Scornec et al., 2025).
These observations align with scenario-based risk assessments that project large declines under continued high emissions, while indicating that stronger mitigation could preserve refuges for the species (Jenouvrier et al., 2020, 2025). Taken together, the evidence supports a clear mechanism: fast-ice loss can cause immediate reproductive failure in bad years, and persistent changes in sea ice conditions are expected to reduce breeding habitat and recruitment across many colonies later this century.
4.1.2 King penguins (sub-Antarctic)
The king penguin (Fig. 2b) is the second-largest penguin species, reaching about 0.9–1.0 m in height and up to 18 kg in mass (see Fig. 2 for size scale). It breeds on sub-Antarctic islands such as South Georgia, Crozet, and Kerguelen, where colonies can number in the hundreds of thousands and the global population exceeds 1 million breeding pairs (Croxall and Prince, 1980; Williams, 1995). During chick rearing, adults often travel hundreds of kilometers to forage near productive frontal systems, especially the Polar Front. A 25-year dataset from Kerguelen shows mean foraging distances of roughly 266 km (with strong interannual variability) and confirms that colder sea surface temperatures and a shallower winter water (WW) layer are associated with higher prey-capture activity, consistent with bottom-up control through prey fields (Brisson-Curadeau et al., 2023).
Recent comparisons also show that neighboring king penguin populations can respond very differently to warming. At Crozet, warmer years were linked to reduced breeding success when productive currents shifted away from the colony and increased foraging constraints during chick rearing. At Kerguelen, year-to-year variation was instead more consistent with changes in prey availability and local conditions, with some evidence that warmer conditions can increase chick survival by reducing thermoregulatory costs in winter (Brisson-Curadeau et al., 2023). Long-term monitoring in the French Southern Territories likewise reports increases at many king penguin sites in Crozet and Kerguelen, while emphasizing that drivers can differ among colonies and remain incompletely explained (Barbraud et al., 2020). In addition, recent observations of king penguins on Elephant Island, including incubation and molting, suggest the potential for further southward range expansion under warming conditions, although successful fledging has not yet been documented there (Borowicz et al., 2020). On land, heat load driven by solar radiation and low wind has also been proposed as a frequent stressor during breeding that can increase the likelihood of reproductive failure on warmer days, although this evidence is currently from a preprint (Noiret et al., 2024).
Although king penguins are currently assessed as Least Concern (BirdLife International, 2023), trends can differ sharply among colonies. For example, on Macquarie Island, chick numbers censused from 2007 to 2020 showed strong interannual variability but an overall decline of about 1 % yr−1 , with lower chick numbers associated with wetter or stormier conditions on land and warmer sea surface temperatures in the foraging region during incubation and early chick rearing (Pascoe et al., 2022). There is also growing evidence for new pressures even in remote sub-Antarctic settings, including documented macroplastic ingestion in the Crozet archipelago (Robin et al., 2024). Together with contrasting responses reported for Crozet versus Kerguelen, this illustrates why climate impacts on king penguins are best interpreted at the colony scale through the mechanisms that affect foraging costs and chick provisioning (Brisson-Curadeau et al., 2023).
4.1.3 Adélie penguins (Antarctic)
The Adélie penguin (Fig. 2c) is a high-latitude breeder whose nesting range is largely restricted to the Antarctic continent and nearby islands. Adélies breed on ice-free ground but depend on the surrounding sea ice to structure access to the ocean and to shape prey fields. Sea ice can therefore influence them through two linked pathways. First, it affects the distance and routes needed to reach foraging grounds. Second, it shapes where predictable prey concentrations form, especially near the sea ice edge and along the continental slope.
Their global population is large, and the species is currently classified as Least Concern (IUCN, 2023). However, long-term trends differ strongly among regions and colonies. For example, a multi-decade synthesis from Cape Royds (Ross Sea) shows that colony size reflects a shifting balance between open-water access, sea ice conditions, and additional pressures including predation and competition, with clear population responses to major episodic events such as iceberg-driven increases in sea ice (Ainley et al., 2024). Recent field surveys north and east of the so-called “Adélie gap” also show that Adélies can remain abundant in parts of the broader Antarctic Peninsula region even while strong declines occur elsewhere, indicating sharp spatial transitions in ecosystem state and habitat suitability (Wethington et al., 2023).
Mechanistic studies help to explain why these contrasts occur. Tracking work from East Antarctica indicates that access to nearby open water is not necessarily the main constraint early in the season. Even when local polynyas or leads are available, incubating adults can preferentially target the more distant sea ice edge and continental slope, where prey availability is more predictable (Michelot et al., 2020). This fits with emerging evidence that the relationship between sea ice and performance is nonlinear. Long-term biologging and energetics analyses link winter sea ice conditions to foraging effort, body condition, and fledging success, with intermediate conditions often associated with better outcomes (Dupuis et al., 2026). At longer timescales, population reconstructions from the Ross Sea region suggest that atmospheric and ocean circulation patterns that affect sea ice and polynyas can leave a strong imprint on Adélie population trajectories, even under periods that were not uniformly warm (Zheng et al., 2022).
Finally, Adélie responses are increasingly documented through changes in timing as well as abundance. A recent landscape-scale camera network shows that breeding phenology in Adélies, together with chinstrap and gentoo penguins, has advanced rapidly over the past decade in the Antarctic Peninsula region, with implications for trophic mismatch and interspecific competition where niches are converging (Juarez Martinez et al., 2026). Taken together, these studies show why Adélie trends cannot be described with a single continent-wide narrative: outcomes depend on how local sea ice conditions, prey fields, and colony-level constraints interact through time.
4.1.4 Gentoo penguins (Antarctic and sub-Antarctic)
Gentoo penguins (Fig. 2d) breed across many sub-Antarctic islands and along the northern Antarctic Peninsula, and they generally use a broader range of habitats than the more ice-dependent Adélies. They breed on ice-free ground, can exploit a variety of prey, and often adjust foraging behavior to local conditions. This flexibility helps to explain why their trends can differ from krill specialists or sea ice obligates, but it does not remove risk.
In the western Antarctic Peninsula region, gentoo populations have increased and expanded southward over recent decades (Hinke et al., 2007; Lynch et al., 2012). Tracking work also shows that gentoo winter habitat use is strongly concentrated in shallow coastal regions along the peninsula, with movements that are not directly determined by sea ice but are still constrained by it (Korczak-Abshire et al., 2021). This matters for conservation because the same coastal corridors are also where human activity and some fisheries overlap.
At the colony scale, gentoo ecology is shaped by both interspecific and intraspecific interactions. For example, at neighboring colonies where chinstraps and gentoos breed sympatrically, tracking and stable isotopes indicate substantial spatial overlap between species but clearer dietary separation, and the strongest competitive signal may occur between nearby colonies of the same species rather than between species (Lee et al., 2021). Such results help to explain why gentoo responses can vary across short distances.
Gentoo colonies are also frequently exposed to direct human disturbance because they often breed at accessible coastal sites visited by tourists. Experiments that simulated realistic visitor behaviors show that an active human presence increases vigilance responses, supporting guidance to keep quiet, move slowly, and increase viewing distance, particularly early in the breeding season (Cajiao et al., 2022). Together with other studies of disturbance and site use, this supports continued management even where gentoo populations are not currently declining (Flynn et al., 2023, 2026). Pollution is an additional concern, including evidence of microplastic exposure in Southern Ocean food webs and seabirds (Kim et al., 2023).
Overall, gentoo penguins illustrate a key point for this synthesis: ecological flexibility can buffer some climate pressures in the short term, but it can also increase exposure to local risks, especially concentrated coastal fisheries, tourism, and disturbance. Interpreting gentoo trends therefore requires attention to local habitat use, prey fields, and human activity, not only broad climate indicators.
4.1.5 Chinstrap penguins (Antarctic and sub-Antarctic)
Chinstrap penguins (Fig. 2e), named for the narrow band under their heads, breed mainly across the Scotia Arc and the Antarctic Peninsula, with major strongholds on the South Sandwich Islands. They are strongly krill dependent, so their population dynamics are closely tied to sea ice conditions and food web change. A global assessment combining satellite imagery, drone surveys, and ground counts estimated about 3.42 million breeding pairs across 375 colonies and found that many colonies with historical benchmarks have declined since the 1980s, while some have increased (Strycker et al., 2020). Regional declines around the northern Antarctic Peninsula are often interpreted as the indirect effects of warming and sea ice change acting through krill availability and foraging conditions, with potential amplification where fisheries overlap with predator foraging grounds (Forcada and Trathan, 2009; Warwick-Evans et al., 2018).
Recent tracking and foraging studies help to make these pathways more explicit. Winter tracking from multiple colonies shows high individual variation and the broad use of ice-free habitats but also suggests that local migration strategies may be linked to local population trends, consistent with a shared winter environmental signal underlying regional change (Hinke et al., 2019). During breeding, oceanographic variability can rapidly alter foraging behavior: coastal downwelling conditions were associated with fewer but longer trips in a local krill fishing area, consistent with reduced prey availability at scales relevant to breeding adults (Lowther et al., 2018). Chinstraps are still listed as Least Concern, but their krill dependence and sensitivity to climate-driven prey fields make them a priority for continued monitoring and ecosystem-based management.
4.1.6 Macaroni penguins (Antarctic and sub-Antarctic)
The macaroni penguin (Fig. 2f) is a crested penguin that breeds mainly on sub-Antarctic islands and remains one of the most abundant penguin species, with the largest share of the global population concentrated at South Georgia and other island groups (Croxall et al., 2002; Ratcliffe et al., 2015; Strycker et al., 2021). Despite high global numbers, many colonies have shown long-term declines, and trends can differ strongly among regions and breeding sites (Croxall et al., 2002; Strycker et al., 2021).
In the Antarctic Peninsula region, macaroni penguins form only a small, peripheral part of the species' range. A recent synthesis of census records across the South Shetland and adjacent island groups estimated about 8053 breeding pairs and suggests a modest increase since the early 1990s, while also highlighting major data gaps in the South Shetlands and South Orkneys, and the need for updated counts at several sites (Hallet and Lynch, 2024). At Elephant Island, a modern census placed present-day abundance in a longer context, reinforcing the fact that local trends in the peninsula region do not necessarily track the larger changes occurring at the main sub-Antarctic strongholds (Strycker et al., 2021).
Work at sub-Antarctic colonies helps to clarify likely pathways linking environmental change to breeding performance. At Marion Island, long-term diet records during chick rearing show that macaroni penguins remained dominated by crustaceans with no clear directional dietary shift over 1994–2018, suggesting that population declines there are unlikely to be explained by simple changes in chick diet composition alone (Dakwa et al., 2021). At the individual scale, biologging and animal-borne video show that foraging effort and efficiency depend on prey type, patch density, and the timing of prey encounters, highlighting how changes in prey fields can increase foraging costs even without an obvious shift in the prey list (Sutton et al., 2021). These sensitivities are relevant to current risk assessments because penguin habitats across the Southern Hemisphere are increasingly exposed to overlapping extreme events, which can compound stress on breeding and foraging conditions (Gimeno et al., 2025).
4.1.7 Southern rockhopper penguins (sub-Antarctic)
Southern rockhoppers (Fig. 2g) are small crested penguins that breed on exposed, rocky coasts of sub-Antarctic islands. Here, “rockhopper penguin” refers to the southern rockhopper penguin (Eudyptes chrysocome) as treated in IUCN and BirdLife assessments (BirdLife International, 2023; IUCN, 2023). Many studies also refer to the “eastern rockhopper” form (E. chrysocome filholi); because taxonomic treatment varies among sources, results are reported by colony region where possible.
Long-term declines have been reported across parts of the range (Croxall et al., 2002), but the drivers and trajectories differ among colonies. A key reason is that southern rockhopper penguins behave like a network of colonies rather than isolated populations. Most birds return to breed near where they were born, but some move between colonies, especially when conditions are poor. Genetic and stable isotope evidence from southern South America suggests that there are two broad regional groups with little mixing between them and that colonies often feed in different areas or rely on different prey (Lois et al., 2020). This means that colonies can respond differently to the same year of ocean conditions, depending on where they forage and how much movement occurs among sites. The same study suggests that dispersal may increase under unfavorable conditions, which can promote the colonization of new sites but does not necessarily offset declines at established colonies.
Tracking and movement studies support the idea that exposure to environmental variability and fisheries risk can change seasonally. For example, birds from Isla Noir used shelf waters during breeding but ranged widely during winter, including offshore basin areas, implying that pressures on survival can occur far from breeding sites (Oehler et al., 2018). Diet studies also show that prey composition can shift in ways that matter for management. Around Campbell Island, stable isotope work indicates that warmer conditions may have enabled the appearance of a commercially important arrow squid in the diet of eastern rockhopper penguins, highlighting how ocean warming and changing prey communities can alter trophic links and potential fisheries overlap (Guímaro et al., 2025).
Together, these findings strengthen the case that population change in rockhopper penguins should be interpreted at the scale of colony networks and ocean regions, not only individual breeding sites, and that climate variability can act through both dispersal behavior and prey field reorganization.
4.1.8 Magellanic penguins (sub-Antarctic)
The Magellanic penguin (Fig. 2h) breeds mainly along the coasts of Argentina and Chile, and on the Falkland (Malvinas) Islands, but it is also occasionally sighted at high latitudes around the South Shetlands and the Antarctic Peninsula (Boersma, 2008). Unlike Antarctic ice-breeding species, Magellanics typically nest in burrows or shaded ground, so their breeding success is strongly shaped by local weather on land as well as prey conditions at sea. Regional declines have been linked to oil pollution, fisheries impacts on key forage fish, and changing ocean conditions (Boersma, 2008; Millones et al., 2021; Nievas El Makte et al., 2021). Recent work also highlights two additional features that matter for conservation planning. First, colonies can shift in space. A newly reported small colony on a sub-Antarctic island (Staten Island) shows that new breeding sites can appear, likely through local dispersal from nearby colonies (Raya Rey et al., 2022). Second, extreme heat can cause direct mortality, not only indirect food web effects. A single-day heat event at Punta Tombo caused substantial adult and chick deaths, showing that hot weather can be an acute, lethal stressor in this species (Holt and Boersma, 2022). Finally, an updated synthesis of breeding colonies along the Argentine coast suggests that, while trends differ among colonies, the overall pattern in recent decades includes redistribution and growth at some sites, with a recent estimate of about 1.28 million breeding pairs by 2024 (Hombre et al., 2026). Together, these results reinforce why Magellanic penguins are a useful ecological bridge between South America and the broader Southern Ocean: they respond to offshore prey fields and human pressures at sea, but they also record land-based climate stress that is likely to intensify with warming.
The conservation status, approximate global population sizes, and principal threats for all eight species are summarized in Table 1. These profiles show that penguin trends are not moving in a single direction, because each species is tied to a different mix of breeding habitat, sea ice conditions, and prey pathways. Where the physical platform for breeding is lost, or where prey access changes through sea ice and ocean front dynamics, demographic signals appear quickly as reduced breeding success, altered phenology, or range shifts. In this sense, the living species complement the fossil and Holocene archives by translating environmental change into measurable biological outcomes that matter for conservation planning and monitoring.
(Trathan et al., 2020; Fretwell, 2024)(Brooks et al., 2018; Tixier et al., 2019)(Iles et al., 2020; Schmidt et al., 2023)(Flynn et al., 2023, 2026; Kim et al., 2023)(Krüger, 2023; Salmerón et al., 2023)(Thiebot and Bost, 2023)(Barrionuevo et al., 2025)(Golubev, 2021; Barrionuevo et al., 2025)Table 1Conservation status, approximate global population size, and principal threats for Antarctic and sub-Antarctic penguin species. Population estimates and conservation status follow the most recent IUCN Red List and BirdLife International assessments (IUCN, 2023; BirdLife International, 2023). Species-specific threat evidence is supported by recent ecological and conservation studies (see citations in table).
4.2 Distribution and habitats
Penguins breed across a broad latitudinal arc that runs from the Antarctic continent to the sub-Antarctic islands and the southern coasts of South America (Fig. 3). Emperor and Adélie penguins are the only species that breed entirely within Antarctica, where breeding success depends on seasonal access to sea ice and nearby open water. King, macaroni, and southern rockhopper penguins breed mainly on sub-Antarctic islands, where they rely on predictable marine feeding zones rather than sea ice platforms. Gentoo and chinstrap penguins span the sub-Antarctic and the northern Antarctic Peninsula, while Magellanic penguins extend the broader system northward into South America and the Falklands. These distributions reflect how penguins partition the Southern Ocean by sea ice conditions, latitude, and prey fields (Williams, 1995; Fraser and Trivelpiece, 1996; Trathan et al., 2007).
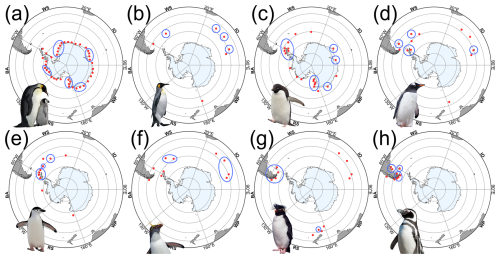
Figure 3Approximate breeding distributions of Antarctic and sub-Antarctic penguins. Maps created by the author in R; distributions adapted from De Roy et al. (2022) and supplemented by published literature (e.g., Fretwell et al., 2012; Lynch and LaRue, 2014; Fretwell and Trathan, 2021). Penguin images are publication-licensed stock illustrations from Canva (Canva for Education), assembled by the author; used under the Canva Content License Agreement (https://www.canva.com/en_in/policies/content-license-agreement/) for online or electronic publication and for non-commercial purposes. Red points mark known breeding sites or colonies, whereas blue circles highlight major population strongholds. Species panels: (a) emperor (A. forsteri), (b) king (A. patagonicus), (c) Adélie (P. adeliae), (d) gentoo (P. papua), (e) chinstrap (P. antarctica), (f) macaroni (E. chrysolophus), (g) southern rockhopper (E. chrysocome), and (h) Magellanic (S. magellanicus). Sector labels correspond to standard Southern Ocean regions following Parkinson (2019): RS = Ross Sea, BA = Bellingshausen–Amundsen Seas, WS = Weddell Sea, IO = Indian Ocean sector, WP = West Pacific sector. The outer ring corresponds to 30° S, with inner concentric rings every 10° of latitude.
Two environmental features are especially important for explaining these patterns. First, sea ice sets both the breeding platform and the travel costs for Antarctic specialists. For emperor penguins, sea ice is a direct requirement because colonies form on fast ice. For Adélie penguins, sea ice mainly acts through access to foraging areas. Too much ice can force longer trips and reduce chick provisioning, while too little ice can remove predictable ice edge habitats and destabilize local food webs. Tracking and diet studies show that Adélies often target the sea ice edge and continental slope even when nearer open water exists, which highlights the fact that prey availability matters as much as simple access (Michelot et al., 2020).
Second, ocean fronts structure the feeding landscape for many sub-Antarctic breeders. The Antarctic Polar Front concentrates productivity and helps to aggregate mid-trophic prey, which is why long foraging trips by king penguins repeatedly align with frontal zones (Trathan et al., 2006). Recent work shows that warming can produce contrasting outcomes across nearby king penguin populations because the same climate forcing can shift productive currents away from one colony while leaving another colony buffered, or even favored, depending on local oceanography (Brisson-Curadeau et al., 2023). This helps to explain why some colonies grow, while others decline even within the same species.
Range shifts already show that penguins respond quickly when these environmental boundaries move. Gentoo penguins have expanded southward in parts of the Antarctic Peninsula region, while some chinstrap populations have declined where krill availability has changed, linking distributional change to prey field reorganization rather than temperature alone (Hinke et al., 2007; Lynch et al., 2012; Trivelpiece et al., 2011). At the same time, Adélie trends differ sharply by region, with stability or growth in parts of East Antarctica contrasting with declines elsewhere, which points to strong regional control by sea ice dynamics and local circulation (Wethington et al., 2023).
Because many penguins nest in dense colonies that are visible from the ground, air, and satellites, their distributions can be mapped and updated more consistently than those of many other marine predators (Fretwell et al., 2012; Fretwell and Trathan, 2021). This makes penguins especially useful for detecting changes in where and when the Southern Ocean can support high predator demand, and it provides a practical baseline for the future projections discussed in Sect. 5.
4.3 Human pressures and efforts
Climate change is not the only driver of change in penguin populations. Penguins also face direct human pressures that can amplify climate-driven risks because they often act on the same bottlenecks: food availability, safe breeding space, and survival during energetically demanding periods.
The most widespread pressure is resource overlap with industrial fisheries. The krill fishery operates in regions where krill is also a keystone prey for several penguin species, creating the potential for localized competition during sensitive breeding stages and in winter foraging areas (Trivelpiece et al., 2011; Pinkerton et al., 2016). The Commission for the Conservation of Antarctic Marine Living Resources (CCAMLR) was created to manage these risks under an ecosystem approach, but the main challenge is spatial and seasonal matching: predators and fisheries concentrate in the same places at the same times, so total catch limits alone may not prevent local depletion.
Tourism and other on-shore activities are increasingly important around the Antarctic Peninsula, where many visits occur in ice-free coastal areas that are also prime penguin nesting habitat. The key issue is not only whether penguins flee but whether repeated disturbance changes vigilance, nest attendance, and the energy budgets of adults during incubation and chick rearing. Field experiments that mimic realistic tourist behavior show that an active human presence increases strong vigilance responses, supporting cautious approach distances and slower, quieter movement, especially early in the breeding season (Cajiao et al., 2022). Broader syntheses of Antarctic tourism emphasize that impacts can be site specific and depend on prior exposure, which argues for careful site management rather than assuming one rule fits all (Huddart and Stott, 2019).
Pollution is another pressure with growing evidence in Antarctica and the sub-Antarctic. Classic cases include oil impacts on Magellanic penguins, but attention is now expanding to plastics, debris, fuel-related contamination around stations, and emerging contaminants that can accumulate through food webs. Along the West Antarctic Peninsula, particle tracking and ocean modeling suggest that debris loads near penguin colonies can be dominated by coastal point sources linked to human activity (tourism, research, and fishing), and that the dominant source varies by region, which helps to prioritize monitoring and mitigation (Gallagher et al., 2024). These patterns also imply that local prevention and cleanup can have outsized benefits compared with assuming that debris is mainly imported from far away. Even when direct mortality is hard to attribute, rising exposure increases the likelihood of sublethal effects (for example, impaired condition or added physiological stress) and may interact with nutritional stress during poor foraging years.
Governance provides both constraints and opportunities for reducing these risks. The Protocol on Environmental Protection to the Antarctic Treaty (Madrid Protocol) sets the core environmental protection framework for activities in Antarctica, while coordination through the Council of Managers of National Antarctic Programs (COMNAP) and SCAR guidance for field practices aims to reduce impacts from science and logistics at sites that also function as long-term biological observatories (Cordonnery, 1998; Tejedo et al., 2012; Rogan-Finnemore, 2018; Hughes et al., 2022).
Long-term penguin monitoring is now carried out by many national Antarctic programs, often with increasing coordination and data sharing. In the United Kingdom, the British Antarctic Survey supports multi-decadal monitoring on the Antarctic Peninsula and South Georgia, including work based from stations such as Rothera and long-running sub-Antarctic field sites (Holdgate, 1965; Cannone et al., 2018). In Australia, the Australian Antarctic Division has sustained long-term Adélie penguin research in East Antarctica, including the Windmill Islands region, alongside broader ecosystem monitoring (Emslie and Woehler, 2005). In the United States, long-term ecological research on the western Antarctic Peninsula has linked penguin population dynamics to sea ice, ocean variability, and food web change through the Palmer Long Term Ecological Research (LTER) program (Smith et al., 1995).
Comparable monitoring and research efforts are also maintained by programs in Chile (e.g., Instituto Antártico Chileno or INACH, and the Chilean Antarctic program in the South Shetlands and Antarctic Peninsula), Argentina (including stations near major colonies such as Esperanza Base in Hope Bay), France (including long-term work in Adélie Land), Germany (with polar infrastructure and environmental monitoring linked to sites such as Neumayer III), New Zealand (Ross Sea research linked to Scott Base), Japan (long-standing Antarctic research including seabird ecology and monitoring), and China (rapidly expanding Antarctic infrastructure, including the newer Qinling station in the Ross Sea sector) (Elliott, 2005; Watanabe, 2014; Richter et al., 2018; Olivares et al., 2019; Rümmler et al., 2021; Libertelli et al., 2024; Paris et al., 2025; You, 2025).
At the same time, governance debates are widening beyond mitigation toward questions of responsibility and representation, including how the interests of non-human residents are considered in Antarctic decision-making (Liebeaux, 2024; Hemmings, 2025). Common infrastructure has also grown around shared observing systems and open repositories, including continent-scale colony databases (e.g., Mapping Application for Penguin Populations and Projected Dynamics, or MAPPPD) and camera network or citizen science platforms used to extend monitoring coverage (Humphries et al., 2017; Freire et al., 2021).
Climate-driven change sets the background trend, but these other pressures often decide whether colonies can cope year to year. Sea ice anomalies and shifting prey fields already narrow the margin for successful breeding and survival; and fisheries overlap, disturbance, and pollution can compound those same constraints by reducing prey access, increasing energetic costs, or adding stress during the most sensitive life stages. Because these drivers can coincide in the same places and seasons, risk is rarely additive in practice: small, repeated impacts can tip outcomes in poor-ice or low-krill years. This is why forward-looking penguin conservation needs to move beyond single-stressor thinking and pair climate-aware projections with locally actionable management, including spatially explicit fishery measures, site-specific visitor and station protocols, and targeted prevention and cleanup around colony hotspots.
Penguins have persisted through large natural shifts in climate and ice over millions of years, but the next century is different in two ways. First, the pace of change is faster than most changes inferred from colony and fossil archives. Second, the main drivers will change at the same time: sea ice, ocean structure, prey availability, and direct human pressures. Future risk therefore depends less on a single factor and more on how these linked pathways reinforce one another.
5.1 Climate and sea ice futures
Future trajectories for Antarctic and sub-Antarctic penguins will be shaped above all by changes in sea ice, ocean fronts, and prey. Sea ice sets the breeding platform for ice-dependent species and also controls feeding conditions by altering the ice edge, polynyas, and distances adults must travel.
Most global climate models project net Antarctic sea ice loss over the 21st century across emissions scenarios, but the rate and regional pattern remain uncertain, especially for summer sea ice (Roach et al., 2020). That uncertainty is important for biology, because penguins respond to local ice conditions at a colony scale rather than to the Antarctic-wide mean. New satellite-based studies now show that colony outcomes can shift sharply between years as fast-ice conditions change, including colony relocations, increased movement, and breeding failures when fast ice breaks out too early (Schmidt and Ballard, 2020; Fretwell et al., 2023; Wienecke et al., 2024). These observations strengthen confidence in the mechanism linking sea ice timing to breeding success, and they also show that extreme low-ice years can cause region-wide failure across multiple colonies (Fretwell et al., 2023).
Sea ice impacts are not limited to emperors. Adélie penguins can also fail when sea ice persists for too long in front of colonies, and blocks access to open water or polynyas during critical breeding stages (Ropert-Coudert et al., 2018). At the same time, fine-scale sea ice products and tracking studies show that penguins do not simply benefit from nearby open water. They often target predictable prey fields at the sea ice edge and along bathymetric features, so a local increase in leads or small polynyas does not necessarily compensate for changes in the broader icescape (Labrousse et al., 2019; Michelot et al., 2020). This helps to explain why responses can diverge between regions and even between nearby colonies.
For sub-Antarctic species, sea ice change matters mainly through ocean structure and prey rather than through breeding habitat. Many projections focus on the position and accessibility of productive frontal zones, especially the Polar Front, which concentrates prey for wide-ranging foragers such as king penguins. However, projecting frontal movement is not straightforward because surface warming can shift sea surface temperature (SST) isotherms without the same shift in the deep dynamic front itself, which complicates habitat projections that rely on SSTs as a proxy (Meijers et al., 2019). This is a key uncertainty to flag clearly: the biological risk is real, but the magnitude and geography of that risk depends on how fronts and prey fields are represented in models.
Across species, a further emerging risk is that climate change is increasing the frequency of extreme events and “bad years”. Those extremes can act as bottlenecks, causing abrupt breeding failures or poor juvenile survival that are not captured well by averages alone (Gimeno et al., 2025; Le Scornec et al., 2025). As a result, future projections are strongest when they combine (i) realistic colony-scale sea ice habitat metrics, (ii) prey field constraints, and (iii) explicit treatment of extremes rather than only mean trends (Labrousse et al., 2023; Jenouvrier et al., 2025).
5.2 Species-specific outlooks
The physical drivers in the previous subsection do not affect all penguins equally. Species differ in where they breed, how far they can forage, and how tightly their breeding success is coupled to fast ice, the ice edge, or frontal prey hotspots. As a result, projected risks are uneven across the Antarctic and sub-Antarctic.
Emperor penguins face the clearest pathway of risk, because breeding requires landfast sea ice that lasts through chick rearing, and this habitat is projected to shrink and become less reliable under intermediate- to high-emissions scenarios (Trathan et al., 2020; Jenouvrier et al., 2025). Recent satellite evidence of widespread breeding failure during extreme low-ice years strengthens confidence that abrupt, colony-level losses can occur when conditions cross a threshold (Fretwell et al., 2023). In this sense, the main uncertainty is not whether emperors are sensitive to fast-ice change but how quickly and where repeated “bad years” will accumulate into sustained population decline.
Adélie penguins are expected to show a mixed future, because they occupy both regions that are warming rapidly and regions that remain strongly ice influenced. In the northern Antarctic Peninsula, continued reduction in winter pack ice and shifts in prey availability are consistent with ongoing declines, whereas parts of East Antarctica and the Ross Sea may remain suitable or even improve in some years, depending on local sea ice and polynya dynamics (Lynch and LaRue, 2014). This makes Adélies a useful indicator of regional change, but it also means continent-wide averages can mask opposing trends.
Gentoo penguins are the most likely “range shifters” among the Antarctic breeders. Their ability to breed on ice-free ground and to forage in coastal habitats has supported the southward expansion on the Antarctic Peninsula, and further expansion is plausible as ice-free habitat increases (Hinke et al., 2007; Lynch et al., 2012). The key constraints are likely to shift from habitat access to local limits, such as competition for nearshore prey, overlap with krill fisheries, and disturbance where human activity concentrates.
For sub-Antarctic species, the dominant uncertainty is how prey fields will reorganize as upper-ocean structure changes. King penguins illustrate this clearly because they rely on predictable access to productive frontal zones. Some projections suggest that changes in frontal accessibility could make certain colonies energetically marginal, but the magnitude and geography of that risk depends on how models represent fronts and prey aggregation (Cristofari et al., 2018; Meijers et al., 2019). Macaroni and southern rockhopper penguins are also exposed to prey field shifts around island shelves, where climate-driven changes in krill and fish can combine with fisheries pressure, producing strong regional contrasts among colonies (Croxall et al., 2002; Bost et al., 2015).
5.3 Fisheries and ecosystem interactions
Antarctic krill fisheries overlap the foraging ranges of several penguin species, especially in the Scotia Sea and around the northern Antarctic Peninsula. Risk is highest when fishing is concentrated near colonies during periods when krill are scarce or shifted away from traditional hotspots, because penguins may need to travel farther and spend more time searching for food (Trivelpiece et al., 2011; Pinkerton et al., 2016). Under these conditions, fishing and climate variability can act together rather than separately. For example, analyses that combine catch records with climate indices and penguin monitoring data show that high catches during warm, low-ice winters are associated with a higher probability of negative population growth in gentoo and chinstrap penguins (Krüger et al., 2021).
A key challenge is scale. Catch limits that look small when averaged over the full krill stock can still be high at the local scale that matters to predators. Long-term monitoring and modeling show reduced penguin performance when local harvest rates exceed thresholds, with effects comparable in magnitude to poor environmental conditions (Watters et al., 2020). This helps to explain why broad statements that the fishery is “precautionary” can be misleading if catches are spatially concentrated.
Recent work also shows that overlap is not fixed. When krill availability is low, penguins can expand their foraging ranges, which increases the chance of entering fishing grounds even inside managed areas (Ratcliffe et al., 2021). Dynamic distribution models based on multi-year acoustic surveys similarly identify persistent krill hotspots at shelf edges, and they highlight where intense fishing activity overlaps foraging chinstrap penguins across years (Freer et al., 2025). These studies point to a practical message for management: the question is not only how much krill is removed but where and when it is removed relative to predator demand.
Food web pressure is also shaped by pollutants that move through the same predator–prey pathway. Penguins can act as sentinels for contamination because many compounds bioaccumulate and can be detected in tissues, eggs, or feathers over time (Ellis et al., 2018). In the Antarctic Peninsula region, emerging organic contaminants have been detected in both chinstrap penguins and their key prey, Antarctic krill, showing that chemical exposure can occur within the same systems that support krill-dependent predators (Motas et al., 2025). Metal exposure provides an additional example of how penguins can reflect regional contamination gradients across the Southern Ocean (Álvarez-Varas et al., 2018). Broader reviews further emphasize that local sources linked to research activity, shipping, and growing visitation can add diesel-related compounds, plastics, and metals to coastal environments, increasing the likelihood of chronic, sublethal stressors that may compound poor foraging years (Puasa et al., 2021).
CCAMLR has the tools to reduce risk through spatially explicit and adaptive management, but effectiveness depends on the timely allocation of catches, the protection of recurring predator–krill hotspots, and monitoring that can detect years when ecosystem conditions are already unfavorable (Constable et al., 2000; Trathan et al., 2015). Ongoing climate change is also altering krill habitat and behavior, which may increase competition between fisheries and krill predators if productive habitat contracts or becomes more variable (Kawaguchi et al., 2024). Figure 4 summarizes how climate variability, krill dynamics, fishing concentration, and additional human pressures such as pollution and tourism can act together to shape risk.
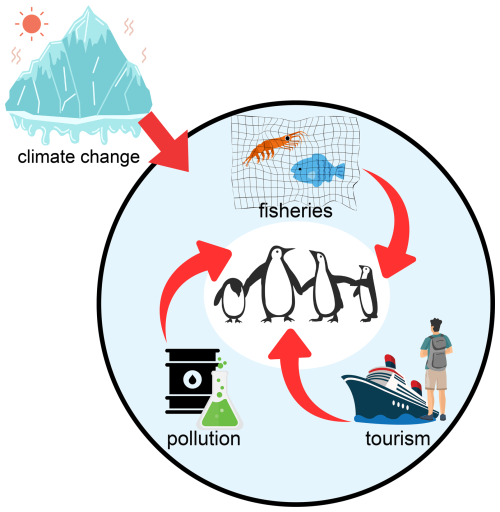
Figure 4Conceptual diagram of direct human pressures on Antarctic and sub-Antarctic penguins. Major pressures include fisheries (competition for krill and fish), pollution (oil spills and contaminants), and tourism (disturbance at breeding sites). Climate change amplifies these stresses by reducing sea ice, shifting prey availability, and increasing the overlap between penguins and human activities. The figure is schematic and intended to summarize interactions rather than provide quantitative estimates.
5.4 Constraints on forecasting penguin futures
Penguin research is extensive, but several bottlenecks still limit confident projections because they sit at the points where population change is decided.
First, the least observed life stages remain the hardest to model. Survival between fledging and recruitment is poorly constrained for many species, even though it strongly controls population growth. Tracking and mark–recapture studies show high variability in juvenile outcomes, and juveniles can use different habitats than adults, sometimes extending beyond areas assumed to capture core foraging needs (Le Scornec et al., 2025).
Second, the non-breeding season is still a weak link for many species. Winter and molt are energetically demanding, but data on where birds forage, what they eat, and how conditions carry over into the next breeding season remain patchy compared with summer chick-rearing datasets. Where winter tracking exists, it often reveals broad dispersal and high individual variation that is difficult to capture with colony-based averages (Hinke et al., 2019).
Third, environmental drivers are frequently represented at the wrong scale for the decisions penguins actually face. Many habitat and demographic models rely on gridded sea ice or ocean products that are too coarse to represent nearshore leads, polynyas, and fast-ice stability at the colony scale. New work using finer sea ice metrics and high-resolution satellite observations shows that these local features can determine access to prey and even trigger breeding failure, highlighting why downscaling and nearshore validation matter (Labrousse et al., 2019, 2023; Wienecke et al., 2024).
Fourth, there are persistent geographic challenges that hinder circum-Antarctic synthesis. Some regions remain under-sampled because of access and logistics, and colony distributions can change our interpretation of “stable” versus “transition” zones. For example, recent surveys north and east of the Antarctic Peninsula document thriving Adélie colonies in areas that contrast with nearby declines and sharpen attention on the so-called Adélie gap (Wethington et al., 2023).
Finally, observation systems still have practical limits. Cloud cover, polar darkness, and variable ice conditions create missingness in remote-sensing time series, while ground-based validation remains uneven across regions. Progress will depend on combining methods rather than relying on any single one: coordinated field counts where feasible; drones for repeatable colony-scale validation; satellite mapping for coverage; and open, reproducible workflows that link population time series to sea ice, ocean structure, prey fields, and human activity (Fretwell and Trathan, 2009; Fretwell et al., 2012; Fretwell and Trathan, 2021; Winterl et al., 2024). This kind of multi-method monitoring has steadily reduced long-standing blind spots in where penguins go, how colonies change from year to year, and how those changes intersect with human activity and governance debates (Blair, 2022).
5.5 Management implications
Because penguin responses vary strongly among regions and species, management works best when it is both precautionary and targeted. A practical starting point is to treat penguins as indicator species that can translate physical change (ice, storms, fronts) and food web change (krill and fish fields) into measurable outcomes such as breeding success, adult foraging effort, and colony persistence. This is already operational in some settings, for example, in the Ross Sea region Marine Protected Area (MPA) where Adélie penguins are used as an indicator of ecosystem structure and function (Smith et al., 2021).
Recent methods make it increasingly feasible to match management actions to the spatial and seasonal scales that matter to penguins. Biologging and habitat models can identify where overlap between predators and fisheries is most likely and can be extended to colonies without direct tracking, helping to prioritize areas for risk management as the krill fishery develops (Trathan et al., 2018). These approaches also highlight the fact that penguin habitat use can shift seasonally, so static boundaries designed around summer distributions may miss a large fraction of the annual cycle (Thiébot et al., 2019). For broader planning, marine Important Bird and Biodiversity Areas provide a transparent way to map high-quality at-sea areas for multiple species, and to evaluate how existing and proposed MPAs align with penguin core habitats (Handley et al., 2021). In parallel, work on movements beyond national jurisdiction shows why governance tools for the high seas can matter for penguins that routinely cross jurisdictional boundaries (Thiébot and Dreyfus, 2021).
Monitoring capacity is also improving rapidly. Remote camera networks can quantify breeding success across large areas with consistent timing and can detect the role of short-lived extreme weather events that may not be visible in seasonal means (Youngflesh et al., 2021). Unstaffed aerial vehicle (UAV) surveys combined with deep learning are becoming useful for colony-scale counts and for mapping local environmental features that guide where to place complementary field sampling (Belyaev et al., 2024). At the same time, new evaluations of guano-based remote sensing show that spatial resolution strongly shapes detection error, reinforcing the need for UAV validation and colony-specific calibration when long Landsat-class archives are used for trend monitoring (Park et al., 2026).
Finally, fishery decisions depend on understanding krill distribution and connectivity at management-relevant scales. Physical ocean modeling shows that krill connectivity can vary sharply in the West Antarctic Peninsula region and that this can intersect with locations where fishing is most intense (Gallagher et al., 2023). Statistical advances are also improving how krill acoustics are modeled in space and time, despite zero inflation and mismatched covariates, which is directly relevant to translating observing systems into decision-ready maps (Amaral et al., 2025). Together, these tools support three near-term priorities: (i) protect key breeding and foraging areas with boundaries and seasons that reflect penguin habitat use, including consideration of mobile or dynamic measures where appropriate; (ii) keep krill catch limits adaptive and spatially distributed to reduce local depletion risk; and (iii) sustain standardized, open monitoring that links colony outcomes to sea ice, prey fields, and human activity. The pathways connecting climate forcing, prey dynamics, and human pressures are summarized in Fig. 5.
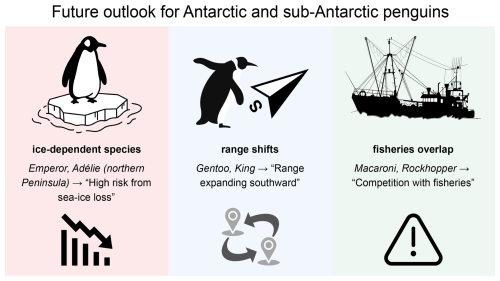
Figure 5Schematic outlook for Antarctic and sub-Antarctic penguins under projected climate and human pressures. Ice-dependent species (emperor, Adélie in the northern peninsula) face high risk from sea ice loss; sub-Antarctic crested species (macaroni, southern rockhopper) face combined risks from changing prey and fisheries overlap. Gentoo and king penguins may shift ranges southward with changing ice and ocean fronts but encounter new constraints where human activity is concentrated. The diagram is conceptual and intended to summarize processes rather than provide quantitative forecasts.
5.6 Outlook for future penguin research
Penguin research has evolved alongside Antarctic science, from expedition-era descriptions to modern monitoring and forecasting (Table 2). Early natural history and expeditions established basic life histories and where penguins breed, whereas the first fossil descriptions revealed that penguins have persisted through profound environmental change over deep time (Ross, 1847; Huxley, 1859). From the mid-20th century onward, long-term colony studies and food web research began to connect breeding outcomes with sea ice, krill, and climate variability, which is what made penguins useful as indicators of ecosystem change (Croxall and Prince, 1980; Barbraud and Weimerskirch, 2001).
Gray (1844); Ross (1847); Huxley (1859); Coues (1872)Levick (1914); Murphy (1947); Marples (1953); Willing (1958)Goldsmith and Sladen (1961); Holdgate (1965); Stonehouse (1967); Oelke and Steiniger (1973); Trivelpiece and Volkman (1979)Croxall and Prince (1980); Volkman et al. (1980); Lishman (1985); Green and Johnstone (1988); Puddicombe and Johnstone (1988)Baroni and Orombelli (1994); Woehler et al. (1994); Williams (1995); Fraser and Trivelpiece (1996); Kirkwood and Robertson (1997)Barbraud and Weimerskirch (2001); Trathan et al. (2006); Emslie and Patterson (2007); Huang et al. (2009)Fretwell et al. (2012); Lynch and LaRue (2014); Trathan et al. (2015); Pertierra et al. (2017); Le Guen et al. (2018); Barreau et al. (2019)Jenouvrier et al. (2021); Blair (2022); Flynn et al. (2023); Fretwell (2024); Lin et al. (2025); Park et al. (2026)Table 2Broad trends in penguin research, showing how dominant questions and methods shifted over time.
Over the last two decades, technology has shifted the scale of what can be observed. Biologging and diet tracers describe where penguins go, what they eat, and how hard they work to raise chicks, while satellite and UAV surveys extend monitoring beyond the few colonies that can be visited regularly (Trathan et al., 2006; Emslie and Patterson, 2007; Fretwell et al., 2012). The result is a shift from asking whether penguins are changing to asking why, where, and under what conditions change accelerates.
Future progress will come less from adding one more case study and more from connecting approaches. The most informative studies will link (i) colony-scale outcomes (breeding success, survival, recruitment), (ii) the local physical setting that penguins actually experience (fast-ice persistence, polynya access, fronts, and prey fields), and (iii) human pressures that operate in the same bottlenecks (especially krill fisheries, disturbance, and pollution). In practice, that means combining continent-wide remote sensing with targeted field validation; and embedding tracking, diet, and demographic data in models that can represent extreme years rather than only long-term averages. This is also where deep-time evidence remains valuable: fossils and geochemical proxies help to define the environmental envelopes penguins have tolerated in the past, while modern monitoring can test whether current rates and combinations of change are pushing populations beyond those envelopes.
Penguins are among the most iconic and visible wildlife of the Antarctic and sub-Antarctic, and their history shows both resilience and vulnerability. Over tens of millions of years, penguins have persisted through major climate transitions, reorganized their ranges as ice advanced and retreated, and diversified into distinct ecological strategies. That deep-time perspective matters because it clarifies the pathways that still govern penguin success today: access to predictable breeding platforms, reliable prey fields shaped by sea ice and ocean fronts, and the ability to buffer occasional poor years.
This overview set out to integrate those perspectives across time. Fossils and paleo proxies show that penguins can survive profound environmental change, but modern observations reveal that today's rapid, human-driven forcing is acting through the same ecological bottlenecks, now compressed into decades and compounded by direct pressures. The result is a widening divergence among species and regions. Some populations are stable or expanding, including southward range shifts by gentoos in parts of the Antarctic Peninsula. Others, especially ice-dependent emperor penguins, are already experiencing breeding failures linked to fast-ice instability, consistent with projected risks under continued warming. Fisheries overlap, pollution, and disturbance can intensify these climate stresses by increasing energetic costs and reducing the margin for successful breeding.
A practical implication is that conservation must match penguin ecology in space and time. This includes protecting recurrent breeding and foraging hotspots, reducing local krill harvest risk during sensitive periods, and designing management that can respond to extreme “bad years” rather than only mean trends. At the same time, recent advances in satellite imagery, UAV surveys, camera networks, biologging, and krill distribution modeling make it increasingly feasible to track penguin populations and their drivers at scales relevant to decision-making. The remaining priority is integration: linking monitoring to demography and prey fields, extending coverage to under-sampled regions and life stages, and making workflows transparent and reusable so that evidence can move efficiently into assessment and management.
In short, penguins have repeatedly demonstrated resilience over deep time, yet their near-future persistence will depend on whether the pace and clustering of modern change outstrips that capacity. By bringing together fossil evidence, present-day monitoring, future scenarios, and research directions, this synthesis highlights both sides of that story. Keeping penguins on the landscape is not only a conservation goal in its own right but also one of the clearest, most measurable ways to track the health and trajectory of the Southern Ocean in the decades ahead.
The ancestral lineage tree shown in the key figure was generated from the morphological dataset of Ksepka et al. (2012) (Dryad repository, https://doi.org/10.5061/dryad.93j174jd). The published Nexus file was uploaded to the Interactive Tree of Life (iTOL; https://itol.embl.de, last access: 16 March 2026) for visualization. The Waimanu tarsometatarsus image included in the key figure is from Skye McDavid (own work; https://commons.wikimedia.org/wiki/File:Waimanu_tarsometatarsus.jpg, last access: 16 March 2026), licensed under Creative Commons Attribution 4.0 International (CC BY 4.0), via Wikimedia Commons (https://creativecommons.org/licenses/by/4.0/, last access: 16 March 2026); this license permits sharing and adaptation for any purpose, including commercial use, provided appropriate attribution is given, a link to the license is provided, and changes are indicated. Penguin images in the key figure are publication-licensed stock illustrations from Canva (Canva for Education), assembled by the author; used under the Canva Content License Agreement (https://www.canva.com/en_in/policies/content-license-agreement/) for online or electronic publication and for non-commercial purposes. All other elements of the key figure were assembled by the author.
The author has declared that there are no competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
Support and encouragement from supervisors and colleagues at the Institut für Geologie und Mineralogie, Universität zu Köln, are gratefully acknowledged. The author is appreciative to Kayshe Joy Fundal Pelingon (ORCID: 0000-0001-5084-7179) for assistance with image editing and figure preparation. The author also thanks Catherine Vase, author of The Penguin Who Wanted to Fly, for inspiring the lasting interest in penguins that led to this study.
This research has been supported by the Deutscher Akademischer Austauschdienst (DAAD; Graduate School Scholarship Programme – ST43) and the Deutsche Forschungsgemeinschaft (DFG; SPP 1158 “Antarktisforschung mit vergleichenden Untersuchungen in arktischen Eisgebieten”).
This paper was edited by Bernhard Diekmann and reviewed by two anonymous referees.
Ainley, D.: The Adélie penguin: bellwether of climate change, Columbia University Press, https://doi.org/10.7312/ainl12306, 2002. a, b
Ainley, D. G., Morandini, V., Barton, K., Lyver, P. O. B., Elrod, M., LaRue, M. A., and Pennycook, J.: Varying population size of the Cape Royds Adélie penguin colony, 1955–2020: a synthesis, Antarct. Sci., 36, 127–140, https://doi.org/10.1017/S0954102024000051, 2024. a
Álvarez-Varas, R., Morales-Moraga, D., González-Acuña, D., Klarian, S. A., and Vianna, J. A.: Mercury exposure in Humboldt (Spheniscus humboldti) and Chinstrap (Pygoscelis antarcticus) penguins throughout the Chilean coast and Antarctica, Arch. Environ. Con. Tox., 75, 75–86, https://doi.org/10.1007/s00244-018-0529-7, 2018. a
Amaral, A. V. R., Sykulski, A. M., Fielding, S., and Cavan, E.: Navigating challenges in spatio-temporal modelling of Antarctic krill abundance: addressing zero-inflated data and misaligned covariates, Spat. Stat., 100937, https://doi.org/10.1016/j.spasta.2025.100937, 2025. a
Barbraud, C. and Weimerskirch, H.: Emperor penguins and climate change, Nature, 411, 183–186, https://doi.org/10.1038/35075554, 2001. a, b
Barbraud, C., Delord, K., Bost, C. A., Chaigne, A., Marteau, C., and Weimerskirch, H.: Population trends of penguins in the French Southern Territories, Polar Biol., 43, 835–850, https://doi.org/10.1007/s00300-020-02691-6, 2020. a
Baroni, C. and Orombelli, G.: Abandoned penguin rookeries as Holocene paleoclimatic indicators in Antarctica, Geology, 22, 23–26, https://doi.org/10.1130/0091-7613(1994)022<0023:APRAHP>2.3.CO;2, 1994. a, b, c, d, e
Barreau, E., Ropert-Coudert, Y., Delord, K., Barbraud, C., and Kato-Ropert, A.: Scale matters: sea ice and breeding success of Adélie penguins, Polar Biol., 42, 1405–1410, https://doi.org/10.1007/s00300-019-02531-2, 2019. a
Barrionuevo, M., Lisovski, S., Morgenthaler, A., Millones, A., and Frere, E.: Different migration strategies under warming ocean anomalies in penguins: a study of spatial, oceanographic, and isotopic niche segregation, J. Ornithol., 1–13, https://doi.org/10.1007/s10336-025-02330-z, 2025. a, b
Bas, M., Tivoli, A. M., i Godino, I. B., Salemme, M., Santiago, F., Belardi, J. B., Borella, F., Vales, D. G., Crespo, E. A., and Cardona, L.: Changing diets over time: knock-on effects of marine megafauna overexploitation on their competitors in the southwestern Atlantic Ocean, Paleobiology, 49, 176–190, https://doi.org/10.1017/pab.2022.19, 2023. a
Belyaev, O., Román, A., Belliure, J., Navarro, G., Barbero, L., and Tovar-Sánchez, A.: Assessing topographic features and population abundance in an Antarctic penguin colony through UAV-based deep-learning models, Int. J. Appl. Earth Obs., 134, 104124, https://doi.org/10.1016/j.jag.2024.104124, 2024. a
BirdLife International: Species factsheets for penguins, http://datazone.birdlife.org (last access: 10 September 2025), 2023. a, b, c, d, e
Blair, J. J.: Tracking penguins, sensing petroleum: “Data gaps” and the politics of marine ecology in the South Atlantic, Environ. Plann. E, 5, 60–80, https://doi.org/10.1177/2514848619882938, 2022. a, b
Boersma, P. D.: Penguins as marine sentinels, Bioscience, 58, 597–607, https://doi.org/10.1641/B580707, 2008. a, b, c, d
Borowicz, A., Forrest, S., Wethington, M., Strycker, N., and Lynch, H. J.: Presence of king penguins (Aptenodytes patagonicus) on Elephant Island provides further evidence of range expansion, Polar Biol., 43, 2117–2120, https://doi.org/10.1007/s00300-020-02760-w, 2020. a
Bost, C.-A., Cotté, C., Bailleul, F., Cherel, Y., Charrassin, J.-B., Guinet, C., Ainley, D. G., and Weimerskirch, H.: Large-scale climatic anomalies affect marine predator foraging behaviour and demography, Nat. Commun., 6, 8220, https://doi.org/10.1038/ncomms9220, 2015. a
Brisson-Curadeau, É., Elliott, K., and Bost, C.-A.: Contrasting bottom-up effects of warming ocean on two king penguin populations, Glob. Change Biol., 29, 998–1008, https://doi.org/10.1111/gcb.16519, 2023. a, b, c, d
Brooks, C. M., Ainley, D. G., Abrams, P. A., Dayton, P. K., Hofman, R. J., Jacquet, J., and Siniff, D. B.: Antarctic fisheries: factor climate change into their management, Nature, 558, 177–180, https://doi.org/10.1038/d41586-018-05372-x, 2018. a
Cajiao, D., Leung, Y. F., Tejedo, P., Barbosa, A., Reck, G., and Benayas, J.: Behavioural responses of two penguin species to human presence at Barrientos Island, a popular tourist site in the Antarctic Peninsula region, Antarct. Sci., 34, 107–119, https://doi.org/10.1017/S0954102021000559, 2022. a, b
Cannone, N., Convey, P., and Malfasi, F.: Antarctic Specially Protected Areas (ASPA): a case study at Rothera Point providing tools and perspectives for the implementation of the ASPA network, Biodivers. Conserv., 27, 2641–2660, https://doi.org/10.1007/s10531-018-1559-1, 2018. a
Clapperton, C. M. and Sugden, D. E.: Holocene glacier fluctuations in South America and Antarctica, Quaternary Sci. Rev., 7, 185–198, https://doi.org/10.1016/0277-3791(88)90005-4, 1988. a, b
Cole, T. L., Zhou, C., Fang, M., Pan, H., Ksepka, D. T., Fiddaman, S. R., Emerling, C. A., Thomas, D. B., Bi, X., Fang, Q., and Ellegaard, M. R.: Genomic insights into the secondary aquatic transition of penguins, Nat. Commun., 13, 3912, https://doi.org/10.1038/s41467-022-31508-9, 2022. a
Constable, A. J., de la Mare, W. K., Agnew, D. J., Everson, I., and Miller, D.: Managing fisheries to conserve the Antarctic marine ecosystem: practical implementation of the Convention on the Conservation of Antarctic Marine Living Resources (CCAMLR), ICES J. Mar. Sci., 57, 778–791, https://doi.org/10.1006/jmsc.2000.0725, 2000. a
Cordonnery, L.: Environmental protection in Antarctica: drawing lessons from the CCAMLR model for the implementation of the Madrid Protocol, Ocean Dev. Int. Law, 29, 125–146, https://doi.org/10.1080/00908329809546120, 1998. a
Coues, E.: Material for a monograph of the Spheniscidae, P. Acad. Nat. Sci. Phila., 24, 170–212, https://www.jstor.org/stable/4624226 (last access: 16 March 2026), 1872. a
Cristofari, R., Liu, X., Bonadonna, F., Cherel, Y., Pistorius, P., Le Maho, Y., Raybaud, V., Stenseth, N. C., Le Bohec, C., and Trucchi, E.: Climate-driven range shifts of the king penguin in a fragmented ecosystem, Nat. Clim. Change, 8, 245–251, https://doi.org/10.1038/s41558-018-0084-2, 2018. a
Croxall, J. P. and Prince, P. A.: Food, feeding ecology and ecological segregation of seabirds at South Georgia, Biol. J. Linn. Soc., 14, 103–131, https://doi.org/10.1111/j.1095-8312.1980.tb00101.x, 1980. a, b, c, d
Croxall, J. P., Trathan, P. N., and Murphy, E. J.: Environmental change and Antarctic seabird populations, Science, 297, 1510–1514, https://doi.org/10.1126/science.1071987, 2002. a, b, c, d, e
Dakwa, F. E., Ryan, P. G., Dyer, B. M., Crawford, R. J. M., Pistorius, P. A., and Makhado, A. B.: Long-term variation in the breeding diets of macaroni and eastern rockhopper penguins at Marion Island (1994–2018), Afr. J. Mar. Sci., 43, 187–199, https://doi.org/10.2989/1814232X.2021.1901248, 2021. a
De Roy, T., Jones, M., and Cornthwaite, J.: Penguins: The Ultimate Guide, Princeton University Press, Princeton, NJ, 2nd Edn., ISBN 9780691233574, https://doi.org/10.2307/j.ctv22rth6w, 2022. a, b
Ducklow, H. W., Baker, K., Martinson, D. G., Quetin, L. B., Ross, R. M., Smith, R. C., Stammerjohn, S. E., Vernet, M., and Fraser, W.: Marine pelagic ecosystems: the west Antarctic Peninsula, Philos. T. Roy. Soc. B, 362, 67–94, https://doi.org/10.1098/rstb.2006.1955, 2007. a
Dupuis, B., Chimienti, M., Angelier, F., Raclot, T., Delord, K., Barbraud, C., Ropert-Coudert, Y., and Kato, A.: Energetics link long-term environmental variations to breeding success in a wild penguin population, J. Animal Ecol., https://doi.org/10.1111/1365-2656.70219, 2026. a
Elliott, C.: Antarctica, Scott Base and its environs, New Zealand Geogr., 61, 68–76, https://doi.org/10.1111/j.1745-7939.2005.00005.x, 2005. a
Ellis, D. S., Cipro, C. V., Ogletree, C. A., Smith, K. E., and Aronson, R. B.: A 50-year retrospective of persistent organic pollutants in the fat and eggs of penguins of the Southern Ocean, Environ. Pollut., 241, 155–163, https://doi.org/10.1016/j.envpol.2018.05.003, 2018. a
Emslie, S. D.: Radiocarbon dates from abandoned penguin colonies in the Antarctic Peninsula region, Antarct. Sci., 13, 289–295, https://doi.org/10.1017/S0954102001000414, 2001. a, b, c
Emslie, S. D.: Ancient Adélie penguin colony revealed by snowmelt at Cape Irizar, Ross Sea, Antarctica, Geology, 49, 145–149, https://doi.org/10.1130/G48230.1, 2021. a, b, c
Emslie, S. D. and Patterson, W. P.: Abrupt recent shift in δ13C and δ15N values in Adélie penguin eggshell in Antarctica, P. Natl. Acad. Sci. USA, 104, 11666–11669, https://doi.org/10.1073/pnas.0608477104, 2007. a, b, c, d, e
Emslie, S. D. and Woehler, E. J.: A 9000-year record of Adélie penguin occupation and diet in the Windmill Islands, East Antarctica, Antarct. Sci., 17, 57–66, https://doi.org/10.1017/S0954102005002427, 2005. a
Emslie, S. D., McKenzie, A., and Patterson, W. P.: The rise and fall of an ancient Adélie penguin supercolony at Cape Adare, Antarctica, Roy. Soc. Open Sci., 5, 172032, https://doi.org/10.1098/rsos.172032, 2018. a
Emslie, S. D., Romero, M., Juáres, M. A., and Argota, M. R.: Holocene occupation history of pygoscelid penguins at Stranger Point, King George (25 de Mayo) Island, northern Antarctic Peninsula, Holocene, 30, 190–196, https://doi.org/10.1177/0959683619875814, 2020. a
Flynn, C. M., Hart, T., Clucas, G. V., and Lynch, H. J.: Penguins in the anthropause: COVID-19 closures drive gentoo penguin movement among breeding colonies, Biol. Conserv., 286, 110318, https://doi.org/10.1016/j.biocon.2023.110318, 2023. a, b, c
Flynn, C. M., Wethington, M., Hilton, M., Clucas, G. V., Hart, T., Humphries, G. R. W., Nichol, C., and Lynch, H. J.: Revisiting “Penguins in the anthropause: Covid-19 closures drive gentoo penguin movement among breeding colonies”, Biol. Conserv., 315, 111715, https://doi.org/10.1016/j.biocon.2026.111715, 2026. a, b
Forcada, J. and Trathan, P. N.: Penguin responses to climate change in the Southern Ocean, Glob. Change Biol., 15, 1618–1630, https://doi.org/10.1111/j.1365-2486.2009.01909.x, 2009. a, b, c
Fraser, W. R. and Trivelpiece, W. Z.: Factors controlling the distribution of seabirds: winter-summer heterogeneity in the distribution of Adélie penguin populations, Foundations for ecological research west of the Antarctic Peninsula, 70, 257–272, https://doi.org/10.1029/AR070p0257, 1996. a, b, c
Freer, J. J., Warwick-Evans, V., Skaret, G., Krafft, B. A., Fielding, S., and Trathan, P. N.: A new dynamic distribution model for Antarctic krill reveals interactions with their environment, predators, and the commercial fishery in the south Scotia Sea region, Limnol. Oceanogr., 70, 833–849, https://doi.org/10.1002/lno.12809, 2025. a
Freire, R., Massaro, M., McDonald, S., Trathan, P., and Nicol, C. J.: A citizen science trial to assess perception of wild penguin welfare, Frontiers in Veterinary Science, 8, 698685, https://doi.org/10.3389/fvets.2021.698685, 2021. a
Fretwell, P. T.: A 6 year assessment of low sea-ice impacts on emperor penguins, Antarct. Sci., 36, 3–5, https://doi.org/10.1017/S0954102024000130, 2024. a, b
Fretwell, P. T. and Trathan, P. N.: Penguins from space: faecal stains reveal the location of emperor penguin colonies, Global Ecol. Biogeogr., 18, 543–552, https://doi.org/10.1111/j.1466-8238.2009.00467.x, 2009. a, b
Fretwell, P. T. and Trathan, P. N.: Discovery of new colonies by Sentinel-2 reveals good and bad news for emperor penguins, Remote Sens. Ecol. Conserv., 7, 139–153, https://doi.org/10.1002/rse2.176, 2021. a, b, c
Fretwell, P. T., LaRue, M. A., Morin, P., Kooyman, G. L., Wienecke, B., Ratcliffe, N., Fox, A. J., Fleming, A. H., Porter, C., and Trathan, P. N.: An emperor penguin population estimate: the first global, synoptic survey of a species from space, PLOS ONE, 7, e33751, https://doi.org/10.1371/journal.pone.0033751, 2012. a, b, c, d, e
Fretwell, P. T., Boutet, A., and Ratcliffe, N.: Record low 2022 Antarctic sea ice led to catastrophic breeding failure of emperor penguins, Commun. Earth Environ., 4, 273, https://doi.org/10.1038/s43247-023-00927-x, 2023. a, b, c, d
Fretwell, P. T., Bamford, C., Skachkova, A., Trathan, P. N., and Forcada, J.: Regional emperor penguin population declines exceed modelled projections, Commun. Earth Environ., 6, 436, https://doi.org/10.1038/s43247-025-02345-7, 2025. a
Gallagher, K. L., Dinniman, M. S., and Lynch, H. J.: Quantifying Antarctic krill connectivity across the West Antarctic Peninsula and its role in large-scale Pygoscelis penguin population dynamics, Sci. Rep., 13, 12072, https://doi.org/10.1038/s41598-023-39105-6, 2023. a
Gallagher, K. L., Cimino, M. A., Dinniman, M. S., and Lynch, H. J.: Quantifying potential marine debris sources and potential threats to penguins on the West Antarctic Peninsula, Environ. Pollut., 347, 123714, https://doi.org/10.1016/j.envpol.2024.123714, 2024. a
Gao, Y., Yang, L., Wang, J., Xie, Z., Wang, Y., and Sun, L.: Penguin colonization following the last glacial-interglacial transition in the Vestfold Hills, East Antarctica, Palaeogeogr. Palaeoclim., 490, 629–639, https://doi.org/10.1016/j.palaeo.2017.11.053, 2018. a
Gimeno, M., Ramírez, F., Coll, M., Chiaradia, A., and Artana, C.: Cumulative extreme events threaten penguin habitats across the Southern Hemisphere, Glob. Change Biol., 31, e70562, https://doi.org/10.1111/gcb.70562, 2025. a, b
Goldsmith, R. and Sladen, W. J. L.: Temperature regulation of some Antarctic penguins, J. Physiol., 157, 251–262, https://doi.org/10.1113/jphysiol.1961.sp006719, 1961. a
Golubev, S.: Seabirds in conditions of local chronic oil pollution, Davis Sea, Antarctica, Birds, 2, 275–284, https://doi.org/10.3390/birds2030020, 2021. a
Gray, G. R.: The Genera of Birds: Comprising Their Generic Characters, a Notice of the Habits of Each Genus, and an Extensive List of Species Referred to Their Several Genera, Longman, Brown, Green, and Longmans, London, https://doi.org/10.5962/bhl.title.60796, 1844. a
Green, K. and Johnstone, G. W.: Changes in the diet of Adélie penguins breeding in east-Antarctica, Wildlife Res., 15, 103–110, https://doi.org/10.1071/WR9880103, 1988. a
Guímaro, H. R., Thompson, D. R., Morrison, K. W., Fragão, J., Matias, R. S., and Xavier, J. C.: Evidence of eastern rockhopper penguin feeding on a key commercial arrow squid species, Polar Biol., 48, 34, https://doi.org/10.1007/s00300-025-03358-w, 2025. a
Hallet, M. and Lynch, H. J.: Update on the abundance and distribution of Macaroni Penguins (Eudyptes chrysolophus) in the Antarctic Peninsula region, Polar Biol., 47, 607–615, https://doi.org/10.1007/s00300-024-03253-w, 2024. a
Handley, J., Rouyer, M. M., Pearmain, E. J., Warwick-Evans, V., Teschke, K., Hinke, J. T., Lynch, H., Emmerson, L., Southwell, C., Griffith, G., and Cárdenas, C. A.: Marine important bird and biodiversity areas for penguins in Antarctica, targets for conservation action, Front. Mar. Sci., 7, 602972, https://doi.org/10.3389/fmars.2020.602972, 2021. a
Hemmings, A. D.: The 47th Antarctic Treaty Consultative Meeting, Milan, Italy in 2025, Polar J.l, https://doi.org/10.1080/2154896X.2025.2545729, 2025. a
Hinke, J. T., Salwicka, K., Trivelpiece, S. G., Watters, G. M., and Trivelpiece, W. Z.: Divergent responses of Pygoscelis penguins reveal a common environmental driver, Oecologia, 153, 845–855, https://doi.org/10.1007/s00442-007-0781-4, 2007. a, b, c, d
Hinke, J. T., Santos, M. M., Korczak-Abshire, M., Milinevsky, G., and Watters, G. M.: Individual variation in migratory movements of chinstrap penguins leads to widespread occupancy of ice-free winter habitats over the continental shelf and deep ocean basins of the Southern Ocean, PLOS ONE, 14, e0226207, https://doi.org/10.1371/journal.pone.0226207, 2019. a, b
Holdgate, M. W.: Biological research by the British Antarctic Survey, Polar Record, 12, 553–573, https://doi.org/10.1017/S0032247400058769, 1965. a, b
Holt, K. A. and Boersma, P. D.: Unprecedented heat mortality of Magellanic penguins, Condor, 124, duab052, https://doi.org/10.1093/ornithapp/duab052, 2022. a
Hombre, J. D., Arias, M., Carrasco, M. F., González, R. A., and Crespo, E. A.: Status and global population trend of the Magellanic penguin Spheniscus magellanicus along the Argentine coast, Sci, Rep,, 16, 3743, https://doi.org/10.1038/s41598-025-33756-3, 2026. a
Huang, T., Sun, L., Wang, Y., Liu, X., and Zhu, R.: Penguin population dynamics for the past 8500 years at Gardner Island, Vestfold Hills, Antarct, Sci,, 21, 571–578, https://doi.org/10.1017/S0954102009990332, 2009. a, b, c, d
Huddart, D. and Stott, T.: Adventure tourism in Antarctica, in: Adventure Tourism: Environmental Impacts and Management, Springer International Publishing, Cham, 11–50, https://doi.org/10.1007/978-3-030-18623-4_2, 2019. a
Hughes, K. A., Santos, M., Caccavo, J. A., Chignell, S. M., Gardiner, N. B., Gilbert, N., Howkins, A., van Vuuren, B. J., Lee, J. R., Liggett, D., Lowther, A., Lynch, H., Quesada, A., Shin, H. C., Soutullo, A., and Terauds, A.: Ant-ICON “Integrated Science to Inform Antarctic and Southern Ocean conservation”: a new SCAR Scientific Research Programme, Antarct. Sci., 34, 446–455, https://doi.org/10.1017/S0954102022000402, 2022. a
Humphries, G. R., Naveen, R., Schwaller, M., Che-Castaldo, J., McDowall, P., Schrimpf, M., and Lynch, H. J.: Mapping application for penguin populations and projected dynamics (MAPPPD): data and tools for dynamic management and decision support, Polar Record, 53, 160–166, https://doi.org/10.1017/S0032247417000055, 2017. a
Huxley, T. H.: On a fossil bird and a fossil cetacean from New Zealand, Q. J. Geol. Soc. Lond., 15, 670–677, https://doi.org/10.1144/GSL.JGS.1859.015.01-02.73, 1859. a, b
Iles, D. T., Lynch, H., Ji, R., Barbraud, C., Delord, K., and Jenouvrier, S.: Sea ice predicts long-term trends in Adélie penguin population growth, but not annual fluctuations: results from a range-wide multiscale analysis, Glob. Change Biol., 26, 3788–3798, https://doi.org/10.1111/gcb.15085, 2020. a
IUCN: The IUCN Red List of Threatened Species, Version 2023-3, https://www.iucnredlist.org (last access: 10 September 2025), 2023. a, b, c, d, e
Jadwiszczak, P.: Eocene penguins of Seymour Island, Antarctica: taxonomy, Polish Polar Research, 3–62, https://journals.pan.pl/Content/110606/PDF/ppr27-003.pdf (last access: 16 March 2026), 2006. a, b
Jenouvrier, S., Holland, M., Iles, D., Labrousse, S., Landrum, L., Garnier, J., Caswell, H., Weimerskirch, H., LaRue, M., Ji, R., and Barbraud, C.: The Paris Agreement objectives will likely halt future declines of emperor penguins, Glob. Change Biol., 26, 1170–1184, https://doi.org/10.1111/gcb.14864, 2020. a
Jenouvrier, S., Che-Castaldo, J., Wolf, S., Holland, M., Labrousse, S., LaRue, M., Wienecke, B., Fretwell, P., Barbraud, C., Greenwald, N., and Stroeve, J.: The call of the emperor penguin: legal responses to species threatened by climate change, Glob. Change Biol., 27, 5008–5029, https://doi.org/10.1111/gcb.15806, 2021. a
Jenouvrier, S., Eparvier, A., Şen, B., Ventura, F., Che-Castaldo, C., Holland, M., Landrum, L., Krumhardt, K., Garnier, J., Delord, K., and Barbraud, C.: Living with uncertainty: using multi-model large ensembles to assess emperor penguin extinction risk for the IUCN Red List, Biol. Conserv., 305, 111037, https://doi.org/10.1016/j.biocon.2025.111037, 2025. a, b, c
Juarez Martinez, I., Kacelnik, A., Jones, F. M., Hinke, J. T., Dunn, M. J., Raya Rey, A., Lynch, H. J., Owen, K., and Hart, T.: Record phenological responses to climate change in three sympatric penguin species, J. Animal Ecol., https://doi.org/10.1111/1365-2656.70201, 2026. a
Kalvakaalva, R., Clucas, G., Herman, R. W., and Polito, M. J.: Late Holocene variation in the hard prey remains and stable isotope values of penguin and seal tissues from the Danger Islands, Antarctica, Polar Biol., 43, 1571–1582, https://doi.org/10.1007/s00300-020-02728-w, 2020. a
Kawaguchi, S., Atkinson, A., Bahlburg, D., Bernard, K. S., Cavan, E. L., Cox, M. J., Hill, S. L., Meyer, B., and Veytia, D.: Climate change impacts on Antarctic krill behaviour and population dynamics, Nat. Rev. Earth Environ., 5, 43–58, https://doi.org/10.1038/s43017-023-00504-y, 2024. a
Kim, Y., Kim, H., Jeong, M.-S., Kim, D., Kim, J., Jung, J., Seo, H.-M., Han, H.-J., Lee, W.-S., and Choi, C.-Y.: Microplastics in gastrointestinal tracts of gentoo penguin (Pygoscelis papua) chicks on King George Island, Antarctica, Sci. Rep., 13, 13016, https://doi.org/10.1038/s41598-023-39844-6, 2023. a, b
Kirkwood, R. and Robertson, G.: Seasonal change in the foraging ecology of emperor penguins on the Mawson Coast, Antarctica, Mar. Ecol. Prog. Ser., 156, 205–223, https://doi.org/10.3354/meps156205, 1997. a
Korczak-Abshire, M., Hinke, J. T., Milinevsky, G., Juáres, M. A., and Watters, G. M.: Coastal regions of the northern Antarctic Peninsula are key for gentoo populations, Biol. Lett., 17, https://doi.org/10.1098/rsbl.2020.0708, 2021. a
Kristan, A. K.: Penguins past and present: trace elements, stable isotopes, and population dynamics in Antarctic and sub-Antarctic penguins and seals, Master's thesis, Louisiana State University and Agricultural and Mechanical College, proQuest Dissertations & Theses Global (Doc ID 2714864648), https://doi.org/10.31390/gradschool_theses.5351, 2021. a
Krüger, L.: Decreasing trends of chinstrap penguin breeding colonies in a region of major and ongoing rapid environmental changes suggest population level vulnerability, Diversity, 15, 327, https://doi.org/10.3390/d15030327, 2023. a
Krüger, L., Huerta, M. F., Santa Cruz, F., and Cárdenas, C. A.: Antarctic krill fishery effects over penguin populations under adverse climate conditions: implications for the management of fishing practices, Ambio, 50, 560–571, https://doi.org/10.1007/s13280-020-01386-w, 2021. a
Ksepka, D. T. and Clarke, J. A.: The basal penguin (Aves: Sphenisciformes) Perudyptes devriesi and a phylogenetic evaluation of the penguin fossil record, B. Am. Mus. Nat. Hist., 1–77, https://doi.org/10.1206/653.1, 2010. a, b, c
Ksepka, D. T., Fordyce, R. E., Ando, T., and Jones, C. M.: Data from: New fossil penguins (Aves, Sphenisciformes) from the Oligocene of New Zealand reveal the skeletal plan of stem penguins, Dryad [data set], https://doi.org/10.5061/dryad.93j174jd, 2012. a
Labrousse, S., Fraser, A. D., Sumner, M., Tamura, T., Pinaud, D., Wienecke, B., Kirkwood, R., Ropert-Coudert, Y., Reisinger, R., Jonsen, I., and Porter-Smith, R.: Dynamic fine-scale sea icescape shapes adult emperor penguin foraging habitat in East Antarctica, Geophys. Res. Lett., 46, 11206–11218, https://doi.org/10.1029/2019GL084347, 2019. a, b
Labrousse, S., Iles, D., Viollat, L., Fretwell, P., Trathan, P. N., Zitterbart, D. P., Jenouvrier, S., and LaRue, M.: Quantifying the causes and consequences of variation in satellite-derived population indices: a case study of emperor penguins, Remote Sens. Ecol. Conserv., 8, 151–165, https://doi.org/10.1002/rse2.233, 2022. a
Labrousse, S., Nerini, D., Fraser, A. D., Salas, L., Sumner, M., Le Manach, F., Jenouvrier, S., Iles, D., and LaRue, M.: Where to live? Landfast sea ice shapes emperor penguin habitat around Antarctica, Sci. Adv., 9, eadg8340, https://doi.org/10.1126/sciadv.adg8340, 2023. a, b
LaRue, M., Iles, D., Labrousse, S., Fretwell, P., Ortega, D., Devane, E., Horstmann, I., Viollat, L., Foster-Dyer, R., Le Bohec, C., Zitterbart, D. P., Houstin, A., Richter, S., Winterl, A., Wienecke, B., Salas, L., Nixon, M., Barbraud, C., Kooyman, G., Ponganis, P., Ainley, D., Trathan, P., and Jenouvrier, S.: Advances in remote sensing of emperor penguins: first multi-year time series documenting trends in the global population, P. Roy. Soc. B, 291, 20232067, https://doi.org/10.1098/rspb.2023.2067, 2024. a
Le Guen, C., Kato, A., Raymond, B., Barbraud, C., Beaulieu, M., Bost, C.-A., Delord, K., MacIntosh, A. J. J., Meyer, X., Raclot, T., Sumner, M., Takahashi, A., Thiebot, J.-B., and Ropert-Coudert, Y.: Reproductive performance and diving behaviour share a common sea-ice concentration optimum in Adélie penguins (Pygoscelis adeliae), Glob. Change Biol., 24, 5304–5317, https://doi.org/10.1111/gcb.14377, 2018. a
Le Scornec, E., Allain, J., Bardon, G., Barracho, T., Chatelain, N., Delord, K., Filippi, D., Ribout, C., Le Bohec, C., and Barbraud, C.: Intrinsic and extrinsic drivers of juvenile survival in emperor penguins, Polar Biol., 48, 41, https://doi.org/10.1007/s00300-025-03369-7, 2025. a, b, c
Lee, W. Y., Park, S., Kim, K. W., Kim, J. H., Gal, J. K., and Chung, H.: Inter-specific and intra-specific competition of two sympatrically breeding seabirds, chinstrap and gentoo penguins, at two neighboring colonies, Animals, 11, 482, https://doi.org/10.3390/ani11020482, 2021. a
Levick, G. M.: Antarctic Penguins: A Study of Their Social Habits, William Heinemann, London, https://doi.org/10.5962/bhl.title.8385, 1914. a
Libertelli, M. M., Orgeira, J. L., and Alvarez, F.: The explorer king: southern report of the king penguin (Aptenodytes patagonicus) in the Antarctic Peninsula, Antarct. Sci., 36, 145–148, https://doi.org/10.1017/S0954102023000354, 2024. a
Liebeaux, J.: “Cats in congress or penguins in parliament”? Towards the just representation of more-than-humans in Antarctic environmental governance, Polar J., 14, 475–492, https://doi.org/10.1080/2154896X.2024.2414643, 2024. a
Lin, H., Cheng, X., Du, J., Kimball, J. S., Yan, Z., Li, T., Li, T., Li, Y., and Chen, Z.: Satellite mapping of emperor penguin habitat dispersal under climate extremes, Remote Sens. Environ., 330, 114984, https://doi.org/10.1016/j.rse.2025.114984, 2025. a, b
Lishman, G. S.: The food and feeding ecology of Adélie penguins (Pygoscelis adeliae) and Chinstrap penguins (P. antarctica) at Signy Island, South Orkney Islands, J. Zoology, 205, 245–263, https://doi.org/10.1111/j.1469-7998.1985.tb03532.x, 1985. a
Lois, N. A., Campagna, L., Balza, U., Polito, M. J., Pütz, K., Vianna, J. A., Morgenthaler, A., Frere, E., Sáenz-Samaniego, R., Raya Rey, A., and Mahler, B.: Metapopulation dynamics and foraging plasticity in a highly vagile seabird, the southern rockhopper penguin, Ecol. Evol., 10, 3346–3355, https://doi.org/10.1002/ece3.6127, 2020. a
Lowther, A. D., Trathan, P., Tarroux, A., Lydersen, C., and Kovacs, K. M.: The relationship between coastal weather and foraging behaviour of chinstrap penguins, Pygoscelis antarctica, ICES J. Mar. Sci.e, 75, 1940–1948, https://doi.org/10.1093/icesjms/fsy061, 2018. a
Lynch, H. J. and LaRue, M. A.: First global census of the Adélie penguin, Auk, 131, 457–466, https://doi.org/10.1642/AUK-14-31.1, 2014. a, b, c
Lynch, H. J., Naveen, R., Trathan, P. N., and Fagan, W. F.: Spatially integrated assessment reveals widespread changes in penguin populations on the Antarctic Peninsula, Ecology, 93, 1367–1377, https://doi.org/10.1890/11-1588.1, 2012. a, b, c, d
Marples, B. J.: Fossil penguins from the mid-Tertiary of Seymour Island, Falkland Islands Dependencies Survey, Scientific Reports No. 5, https://nora.nerc.ac.uk/id/eprint/511133/1/Fossil%20penguins%20from%20the%20mid-Tertiary%20of%20Seymour%20Island%20-%20FIDS%20Scientific%20Report%205.pdf (last access: 16 March 2026), 1953. a, b, c
Martin, S.: Penguin, Reaktion Books, ISBN-13 9781861893765, ISBN-10 1861893760, 2009. a, b, c
Mayr, G., De Pietri, V. L., Love, L., Mannering, A. A., Bevitt, J. J., and Scofield, R. P.: First complete wing of a stem group sphenisciform from the Paleocene of New Zealand sheds light on the evolution of the penguin flipper, Diversity, 12, 46, https://doi.org/10.3390/d12020046, 2020. a
Mayr, G., De Pietri, V. L., Proffitt, J., Blokland, J. C., Clarke, J. A., Love, L., Mannering, A. A., Crouch, E. M., Reid, C., and Scofield, R. P.: Multiple exceptionally preserved fossils from the Paleocene Waipara Greensand inform the diversity of the oldest stem group Sphenisciformes and the formation of their diving adaptations, Zool. J. Linn. Soc., 204, zlaf080, https://doi.org/10.1093/zoolinnean/zlaf080, 2025. a
Meijers, A. J., Meredith, M. P., Murphy, E. J., Chambers, D. P., Belchier, M., and Young, E. F.: The role of ocean dynamics in king penguin range estimation, Nat. Clim. Change, 9, 120–121, https://doi.org/10.1038/s41558-018-0388-2, 2019. a, b
Michelot, C., Kato, A., Raclot, T., Shiomi, K., Goulet, P., Bustamante, P., and Ropert-Coudert, Y.: Sea-ice edge is more important than closer open water access for foraging Adélie penguins: evidence from two colonies, Mar. Ecol. Prog. Ser., 640, 215–230, https://doi.org/10.3354/meps13289, 2020. a, b, c
Michelson, C. I., Polito, M. J., Wunder, M. B., Emslie, S. D., McCarthy, M. D., Patterson, W. P., and McMahon, K. W.: Holocene climate change shifted Southern Ocean biogeochemical cycling and predator trophic dynamics, Limnol. Oceanogr., 68, 2642–2653, https://doi.org/10.1002/lno.12446, 2023. a
Millones, A., Morgenthaler, A., Gandini, P., and Frere, E.: Population numbers of the Magellanic penguin along its central-southern distribution in Argentina: an update after 25 years, Waterbirds, 44, 499–508, https://doi.org/10.1675/063.044.0411, 2021. a
Motas, M., Jerez-Rodríguez, S., Veiga-del Baño, J. M., Ramos, J. J., Oliva, J., Cámara, M. Á., Andreo-Martínez, P., and Corsolini, S.: Emerging pollutants in chinstrap penguins and krill from Deception Island (South Shetland Islands, Antarctica), Toxics, 13, 549, https://doi.org/10.3390/toxics13070549, 2025. a
Murphy, R. C.: A new zonal race of the gentoo penguin, Auk, 64, 454–455, 1947. a
Nievas El Makte, M. L., Polifroni, R., Sepúlveda, M. A., and Fazio, A.: Petroleum hydrocarbons in Atlantic Coastal Patagonia, in: Anthropogenic Pollution of Aquatic Ecosystems, Springer International Publishing, Cham, 325–352, https://doi.org/10.1007/978-3-030-75602-4_15, 2021. a
Noiret, A., Lewden, A., Lemonnier, C., Bocquet, C., Montblanc, M., Bertile, F., Hoareau, M., Marcon, E., Robin, J.-P., Bize, P., and Viblanc, V. A.: Mind the polar sun: solar radiations trigger frequent heat stress in breeding king penguins, despite relatively cool air temperatures, bioRxiv [preprint], https://doi.org/10.1101/2024.09.09.611977, 2024. a
Oehler, D. A., Marin, M., Kusch, A., Danielle, L., Weakley, L. A., and Fry, W. R.: Foraging ranges in southern rockhopper penguins (Eudyptes chrysocome chrysocome) on Isla Noir, Chile, International Journal of Avian & Wildlife Biology, 3, 320–325, https://doi.org/10.15406/ijawb.2018.03.00109, 2018. a
Oelke, H. and Steiniger, F.: Salmonella in Adélie penguins (Pygoscelis adeliae) and south polar skuas (Catharacta maccormicki) on Ross Island, Antarctica, Avian Dis., 568–573, https://doi.org/10.2307/1589155, 1973. a
Olivares, F., Tapia, R., Gálvez, C., Meza, F., Barriga, G. P., Borras-Chavez, R., Mena-Vasquez, J., Medina, R. A., and Neira, V.: Novel penguin avian avulaviruses 17, 18 and 19 are widely distributed in the Antarctic Peninsula, Transbound. Emerg. Dis., 66, 2227–2232, https://doi.org/10.1111/tbed.13309, 2019. a
Paris, J. R., Nitta Fernandes, F. A., Pirri, F., Greco, S., Gerdol, M., Pallavicini, A., Benoiste, M., Cornec, C., Zane, L., Haas, B., and Le Bohec, C.: Gene expression shifts in Emperor penguin adaptation to the extreme Antarctic environment, Mol. Ecol., 34, e17552, https://doi.org/10.1111/mec.17552, 2025. a
Park, J., Hyun, C. U., Kim, Y., Jeong, Y., Yoon, J. E., Guha, S., Kim, J. U., Lee, S. H., Kim, J. H., and Kim, H. C.: Spatial resolution impact assessment for long-term population monitoring of Adélie penguin guano, New Zeal. J. Geol. Geop., 69, e70027, https://doi.org/10.1002/jgo2.70027, 2026. a, b
Parkinson, C. L.: A 40-y record reveals gradual Antarctic sea ice increases followed by decreases at rates far exceeding the rates seen in the Arctic, P. Natl. Acad. Sci. USA, 116, 14414–14423, https://doi.org/10.1073/pnas.1906556116, 2019. a
Pascoe, P., Raymond, B., Carmichael, N., and McInnes, J.: The current trajectory of king penguin (Aptenodytes patagonicus) chick numbers on Macquarie Island in relation to environmental conditions, ICES J. Mar. Sci., 79, 2084–2092, https://doi.org/10.1093/icesjms/fsac139, 2022. a
Pelegrín, J. S. and Acosta Hospitaleche, C.: Evolutionary and biogeographical history of penguins (Sphenisciformes): review of the dispersal patterns and adaptations in a geologic and paleoecological context, Diversity, 14, 255, https://doi.org/10.3390/d14040255, 2022. a
Pertierra, L. R., Hughes, K. A., Vega, M. G., and Olalla-Tarraga, M. A.: High resolution spatial mapping of human footprint across Antarctica and its implications for the strategic conservation of avifauna, PLoS ONE, 12, e0168280, https://doi.org/10.1371/journal.pone.0168280, 2017. a
Pinkerton, M. H., Lyver, P. O. B., Stevens, D. W., Forman, J., Eisert, R., and Mormede, S.: Increases in Adélie penguins in the Ross Sea: Could the fishery for Antarctic toothfish be responsible?, Ecol. Model., 337, 262–271, https://doi.org/10.1016/j.ecolmodel.2016.07.007, 2016. a, b
Puasa, N. A., Zulkharnain, A., Verasoundarapandian, G., Wong, C. Y., Zahri, K. N. M., Merican, F., Shaharuddin, N. A., Gomez-Fuentes, C., and Ahmad, S. A.: Effects of diesel, heavy metals and plastics pollution on penguins in Antarctica: a review, Animals, 11, 2505, https://doi.org/10.3390/ani11092505, 2021. a
Puddicombe, R. A. and Johnstone, G. W.: The breeding season diet of Adélie penguins at the Vestfold Hills, East Antarctica, Hydrobiologia, 165, 239–253, https://doi.org/10.1007/BF00025593, 1988. a
Ratcliffe, N., Hill, S. L., Staniland, I. J., Brown, R., Adlard, S., Horswill, C., and Trathan, P. N.: Do krill fisheries compete with macaroni penguins? Spatial overlap in prey consumption and catches during winter, Divers. Distrib., 21, 1339–1348, https://doi.org/10.1111/ddi.12366, 2015. a
Ratcliffe, N., Deagle, B., Love, K., Polanowski, A., Fielding, S., Wood, A. G., Hill, S., Grant, S., Belchier, M., Fleming, A., and Hall, J.: Changes in prey fields increase the potential for spatial overlap between gentoo penguins and a krill fishery within a marine protected area, Divers. Distrib., 27, 552–563, https://doi.org/10.1111/ddi.13216, 2021. a
Raya Rey, A., Balza, U., Domato, I., and Zunino, F.: New Magellanic penguin Spheniscus magellanicus colony in a subantarctic island, Polar Biol., 45, 1553–1558, https://doi.org/10.1007/s00300-022-03093-6, 2022. a
Richter, S., Gerum, R. C., Schneider, W., Fabry, B., Le Bohec, C., and Zitterbart, D. P.: A remote-controlled observatory for behavioural and ecological research: a case study on emperor penguins, Methods Ecol. Evol., 9, 1168–1178, https://doi.org/10.1111/2041-210X.12971, 2018. a
Ritchie, P. A., Millar, C. D., Gibb, G. C., Baroni, C., and Lambert, D. M.: Ancient DNA enables timing of the Pleistocene origin and Holocene expansion of two Adélie penguin lineages in Antarctica, Mol. Biol. Evol., 21, 240–248, https://doi.org/10.1093/molbev/msh012, 2004. a, b
Roach, L. A., Dörr, J., Holmes, C. R., Massonnet, F., Blockley, E. W., Notz, D., Rackow, T., Raphael, M. N., O'Farrell, S. P., Bailey, D. A., and Bitz, C. M.: Antarctic sea ice area in CMIP6, Geophys. Res. Lett., 47, e2019GL086729, https://doi.org/10.1029/2019GL086729, 2020. a
Robin, J.-P., Bardon, G., Bertile, F., Carette, P., Cillard, A., Lenourry, L., and Le Bohec, C.: When the King penguin meets macroplastics: a first case reported in the Crozet archipelago, Southern Indian Ocean, Mar. Pollut. Bull., 209, 117093, https://doi.org/10.1016/j.marpolbul.2024.117093, 2024. a
Rogan-Finnemore, M.: Council of Managers of National Antarctic Programs (COMNAP): Annual General Meeting (AGM) XXX & Symposium, Polar J., 8, 417–419, https://doi.org/10.1080/2154896X.2018.1541835, 2018. a
Ropert-Coudert, Y., Kato, A., Shiomi, K., Barbraud, C., Angelier, F., Delord, K., Poupart, T., Koubbi, P., and Raclot, T.: Two recent massive breeding failures in an Adélie penguin colony call for the creation of a marine protected area in D'Urville Sea/Mertz, Front. Mar. Sci., 5, 264, https://doi.org/10.3389/fmars.2018.00264, 2018. a
Ross, J. C.: A Voyage of Discovery and Research in the Southern and Antarctic Regions, During the Years 1839–43, John Murray, London, https://doi.org/10.5962/bhl.title.98449, 1847. a, b
Rümmler, M.-C., Esefeld, J., Pfeifer, C., and Mustafa, O.: Effects of UAV overflight height, UAV type, and season on the behaviour of emperor penguin adults and chicks, Remote Sens. Appl., 23, 100558, https://doi.org/10.1016/j.rsase.2021.100558, 2021. a
Salmerón, N., Belle, S., Cruz, F. S., Alegria, N., Finger, J. V. G., Corá, D. H., Petry, M. V., Hernández, C., Cárdenas, C. A., and Krüger, L.: Contrasting environmental conditions precluded lower availability of Antarctic krill affecting breeding chinstrap penguins in the Antarctic Peninsula, Sci. Rep., 13, 5265, https://doi.org/10.1038/s41598-023-32352-7, 2023. a
Schmidt, A. E. and Ballard, G.: Significant chick loss after early fast ice breakup at a high-latitude emperor penguin colony, Antarct. Sci., 32, 180–185, https://doi.org/10.1017/S0954102020000048, 2020. a, b
Schmidt, A. E., Lescroël, A., Lisovski, S., Elrod, M., Jongsomjit, D., Dugger, K. M., and Ballard, G.: Sea ice concentration decline in an important Adélie penguin molt area, P. Natl. Acad. Sci. USA, 120, e2306840120, https://doi.org/10.1073/pnas.2306840120, 2023. a
Slack, K. E., Jones, C. M., Ando, T., Harrison, G. L., Fordyce, R. E., Arnason, U., and Penny, D.: Early penguin fossils, plus mitochondrial genomes, calibrate avian evolution, Mol. Biol. Evol., 23, 1144–1155, https://doi.org/10.1093/molbev/msj124, 2006. a
Smith, R. C., Baker, K. S., Fraser, W. R., Hofmann, E. E., Karl, D. M., Klinck, J. M., Quetin, L. B., Ross, R. M., and Trivelpiece, W. Z.: The Palmer LTER: a long-term ecological research program at Palmer Station, Antarctica, Oceanography, 8, 77–86, https://doi.org/10.5670/oceanog.1995.01, 1995. a
Smith, W. O., Ainley, D. G., Heywood, K. J., and Ballard, G.: New technologies aid understanding of the factors affecting Adélie penguin foraging, Oceanography, 34, 26–27, https://doi.org/10.5670/oceanog.2021.supplement.02-10, 2021. a
Stilwell, J. and Long, J.: Frozen in time: prehistoric life in Antarctica, Csiro Publishing, ISBN-13 9780643096356, ISBN-10 0643096353, 2011. a, b, c
Stonehouse, B.: The general biology and thermal balances of penguins, Adv. Ecol. Res., 4, 131–196, https://doi.org/10.1016/S0065-2504(08)60321-9, 1967. a
Strugnell, J. M., McGregor, H. V., Wilson, N. G., Meredith, K. T., Chown, S. L., Lau, S. C., Robinson, S. A., and Saunders, K. M.: Emerging biological archives can reveal ecological and climatic change in Antarctica, Glob. Change Biol., 28, 6483–6508, https://doi.org/10.1111/gcb.16356, 2022. a
Strycker, N., Wethington, M., Borowicz, A., Forrest, S., Witharana, C., Hart, T., and Lynch, H. J.: A global population assessment of the Chinstrap penguin (Pygoscelis antarctica), Sci. Rep., 10, 19474, https://doi.org/10.1038/s41598-020-76479-3, 2020. a
Strycker, N., Borowicz, A., Wethington, M., Forrest, S., Shah, V., Liu, Y., Singh, H., and Lynch, H. J.: Fifty-year change in penguin abundance on Elephant Island, South Shetland Islands, Antarctica: results of the 2019–20 census, Polar Biol., 44, 45–56, https://doi.org/10.1007/s00300-020-02774-4, 2021. a, b, c
Sun, L., Xie, Z., and Zhao, J.: A 3,000-year record of penguin populations, Nature, 407, 858–858, https://doi.org/10.1038/35038163, 2000. a, b, c, d
Sutton, G. J., Bost, C. A., Kouzani, A. Z., Adams, S. D., Mitchell, K., and Arnould, J. P.: Fine-scale foraging effort and efficiency of Macaroni penguins is influenced by prey type, patch density and temporal dynamics, Mar. Biol., 168, 3, https://doi.org/10.1007/s00227-020-03811-w, 2021. a
Tejedo, P., Pertierra, L. R., Benayas, J., Convey, P., Justel, A., and Quesada, A.: Trampling on maritime Antarctica: can soil ecosystems be effectively protected through existing codes of conduct?, Polar Res., 31, 10888, https://doi.org/10.3402/polar.v31i0.10888, 2012. a
Thiebot, J.-B. and Bost, C.-A.: Exploitation of distant Antarctic habitats by juvenile macaroni penguins: conservation implications, Research Square [preprint], https://doi.org/10.21203/rs.3.rs-3035279/v1, 2023. a
Thiébot, J.-B. and Dreyfus, M.: Protecting marine biodiversity beyond national jurisdiction: a penguins' perspective, Mar. Policy, 131, 104640, https://doi.org/10.1016/j.marpol.2021.104640, 2021. a
Thiébot, J.-B., Ropert-Coudert, Y., Raclot, T., Poupart, T., Kato, A., and Takahashi, A.: Adélie penguins' extensive seasonal migration supports dynamic Marine Protected Area planning in Antarctica, Mar. Policy, 109, 103692, https://doi.org/10.1016/j.marpol.2019.103692, 2019. a
Tixier, P., Giménez, J., Reisinger, R. R., Méndez-Fernandez, P., Arnould, J. P. Y., Cherel, Y., and Guinet, C.: Importance of toothfish in the diet of generalist subantarctic killer whales: implications for fisheries interactions, Mar. Ecol. Prog. Ser., 613, 197–210, https://doi.org/10.3354/meps12894, 2019. a
Trathan, P. N., Green, C., Tanton, J., Peat, H., Poncet, J., and Morton, A.: Foraging dynamics of macaroni penguins Eudyptes chrysolophus at South Georgia during brood-guard, Mar. Ecol. Prog. Ser., 323, 239–251, https://doi.org/10.3354/meps323239, 2006. a, b, c, d
Trathan, P. N., Forcada, J., and Murphy, E. J.: Environmental forcing and Southern Ocean marine predator populations: effects of climate change and variability, Philos. T. Roy. Soc. B, 362, 2351–2365, https://doi.org/10.1098/rstb.2006.1953, 2007. a
Trathan, P. N., García-Borboroglu, P., Boersma, D., Bost, C.-A., Crawford, R. J. M., Crossin, G. T., Cuthbert, R. J., Dann, P., Davis, L. S., De La Puente, S., Ellenberg, U., Lynch, H. J., Mattern, T., Pütz, K., Seddon, P. J., Trivelpiece, W., and Wienecke, B.: Pollution, habitat loss, fishing, and climate change as critical threats to penguins, Conserv. Biol., 29, 31–41, https://doi.org/10.1111/cobi.12349, 2015. a, b
Trathan, P. N., Warwick-Evans, V., Hinke, J. T., Young, E. F., Murphy, E. J., Carneiro, A. P. B., Dias, M. P., Kovacs, K. M., Lowther, A. D., Godø, O. R., and Kokubun, N.: Managing fishery development in sensitive ecosystems: identifying penguin habitat use to direct management in Antarctica, Ecosphere, 9, e02392, https://doi.org/10.1002/ecs2.2392, 2018. a
Trathan, P. N., Wienecke, B., Barbraud, C., Jenouvrier, S., Kooyman, G., Le Bohec, C., Ainley, D. G., Ancel, A., Zitterbart, D. P., Chown, S. L., and LaRue, M.: The emperor penguin-Vulnerable to projected rates of warming and sea ice loss, Biol. Conserv., 241, 108216, https://doi.org/10.1016/j.biocon.2019.108216, 2020. a, b, c, d, e
Trivelpiece, W. and Volkman, N. J.: Nest-site competition between Adélie and Chinstrap penguins: an ecological interpretation, Auk, 96, 675–681, https://academic.oup.com/auk/article-abstract/96/4/675/5184822 (last access: 16 March 2026), 1979. a
Trivelpiece, W. Z., Hinke, J. T., Miller, A. K., Reiss, C. S., Trivelpiece, S. G., and Watters, G. M.: Variability in krill biomass links harvesting and climate warming to penguin population changes in Antarctica, P. Natl. Acad. Sci. USA, 108, 7625–7628, https://doi.org/10.1073/pnas.1016560108, 2011. a, b, c
Vianna, J. A., Fernandes, F. A., Frugone, M. J., Figueiró, H. V., Pertierra, L. R., Noll, D., Bi, K., Wang-Claypool, C. Y., Lowther, A., Parker, P., and Le Bohec, C.: Genome-wide analyses reveal drivers of penguin diversification, P. Natl. Acad. Sci. USA, 117, 22303–22310, https://doi.org/10.1073/pnas.2006659117, 2020. a
Volkman, N. J., Presler, P., and Trivelpiece, W.: Diets of pygoscelid penguins at King George Island, Antarctica, Condor, 82, 373–378, https://doi.org/10.2307/1367558, 1980. a
Warwick-Evans, V., Ratcliffe, N., Lowther, A. D., Manco, F., Ireland, L., Clewlow, H. L., and Trathan, P. N.: Using habitat models for chinstrap penguins Pygoscelis antarctica to advise krill fisheries management during the penguin breeding season, Divers. Distrib., 24, 1756–1771, https://doi.org/10.1111/ddi.12817, 2018. a
Watanabe, K.: The Japanese Antarctic Research Expedition in progress and its organization, Journal of Black Sea/Mediterranean Environment, 20, 78–91, 2014. a
Watters, G. M., Hinke, J. T., and Reiss, C. S.: Long-term observations from Antarctica demonstrate that mismatched scales of fisheries management and predator-prey interaction lead to erroneous conclusions about precaution, Sci. Rep., 10, 2314, https://doi.org/10.1038/s41598-020-59223-9, 2020. a
Wethington, M., Flynn, C., Borowicz, A., and Lynch, H. J.: Adélie penguins north and east of the “Adélie gap” continue to thrive in the face of dramatic declines elsewhere in the Antarctic Peninsula region, Sci. Rep., 13, 2525, https://doi.org/10.1038/s41598-023-29465-4, 2023. a, b, c
Wienecke, B., Lieser, J. L., McInnes, J. C., and Barrington, J. H.: Fast ice variability in East Antarctica: observed repercussions for emperor penguins, Endangered Species Res., 55, 1–19, https://doi.org/10.3354/esr01355, 2024. a, b
Williams, T. D.: The Penguins: Spheniscidae, Oxford University Press, Oxford, ISBN-13 9780198546672, ISBN-10 019854667X, https://documentatiecentrum.watlab.be/imis.php?module=ref&refid=21395 (last access: 16 March 2026), 1995. a, b, c, d, e
Willing, R. L.: Australian discoveries of emperor penguin rookeries in Antarctica during 1954–57, Nature, 182, 1393–1394, https://doi.org/10.1038/1821393a0, 1958. a
Wing, S. R., Wing, L. C., O'Connell-Milne, S. A., Barr, D., Stokes, D., Genovese, S., and Leichter, J. J.: Penguins and seals transport limiting nutrients between offshore pelagic and coastal regions of Antarctica under changing sea ice, Ecosystems, 24, 1203–1221, https://doi.org/10.1007/s10021-020-00578-5, 2021. a
Winterl, A., Richter, S., Houstin, A., Barracho, T., Boureau, M., Cornec, C., Couet, D., Cristofari, R., Eiselt, C., Fabry, B., Krellenstein, A., Mark, C., Mainka, A., Ménard, D., Morinay, J., Pottier, S., Schloesing, E., Le Bohec, C., and Zitterbart, D. P.: Remote sensing of emperor penguin abundance and breeding success, Nat. Commun., 15, 4419, https://doi.org/10.1038/s41467-024-48239-8, 2024. a, b
Woehler, E. J., Penney, R. L., Creet, S. M., and Burton, H. R.: Impacts of human visitors on breeding success and long-term population trends in Adélie penguins at Casey, Antarctica, Polar Biol., 14, 269–274, https://doi.org/10.1007/BF00239175, 1994. a
Wood, J. R., Zhou, C., Cole, T. L., Coleman, M., Anderson, D. P., Lyver, P. O. B., Tan, S., Xiang, X., Long, X., Luo, S., and Lou, M.: Sedimentary DNA insights into Holocene Adélie penguin (Pygoscelis adeliae) populations and ecology in the Ross Sea, Antarctica, Nat. Commun., 16, 1798, https://doi.org/10.1038/s41467-025-56925-4, 2025. a
WWF: Penguins, https://www.worldwildlife.org/species/penguin (last access: 10 February 2026), 2026. a
Xia, B.-Y., Pei, J.-L., and Li, Q.-G.: A new penguin fossil from Seymour Island and reassessment of taxonomy and diversity of Eocene Antarctic penguins, Palaeoworld, 33, 1668–1680, https://doi.org/10.1016/j.palwor.2024.04.007, 2024. a, b
Xu, Q.-B., Gao, Y.-S., Yang, L.-J., Yang, W.-Q., Chu, Z.-D., Wang, Y.-H., Sun, L.-G., and Xie, Z.-Q.: Abandonment of penguin subcolonies in the late nineteenth century on Inexpressible Island, Antarctica, J. Geophys. Res.-Biogeo., 125, e2020JG006080, https://doi.org/10.1029/2020JG006080, 2020. a
You, X.: China's Antarctic research drive offers hope for scientists, Nature, 643, 31, https://doi.org/10.1038/d41586-025-02157-x, 2025. a
Younger, J. L., Clucas, G. V., Kooyman, G., Wienecke, B., Rogers, A. D., Trathan, P. N., Hart, T., and Miller, K. J.: Too much of a good thing: sea ice extent may have forced emperor penguins into refugia during the last glacial maximum, Glob. Change Biol., 21, 2215–2226, https://doi.org/10.1111/gcb.12882, 2015. a
Youngflesh, C., Jones, F. M., Lynch, H. J., Arthur, J., Ročkaiová, Z., Torsey, H. R., and Hart, T.: Large-scale assessment of intra- and inter-annual breeding success using a remote camera network, Remote Sens. Ecol. Conserv., 7, 97–108, https://doi.org/10.1002/rse2.171, 2021. a
Zheng, Z., Nie, Y., Chen, X., Jin, J., Chen, Q., and Liu, X.: Historical population dynamics of the Adélie penguin in response to atmospheric-ocean circulation patterns at Beaufort Island, Ross Sea, Antarctica, Global Planet. Change, 216, 103892, https://doi.org/10.1016/j.gloplacha.2022.103892, 2022. a
- Abstract
- Introduction
- Approach and sources
- Past: penguins as archives of deep time and past environments
- Present: penguins today
- Future: risks, projections, and research challenges
- Concluding remarks
- Data availability
- Competing interests
- Disclaimer
- Acknowledgements
- Financial support
- Review statement
- References
- Abstract
- Introduction
- Approach and sources
- Past: penguins as archives of deep time and past environments
- Present: penguins today
- Future: risks, projections, and research challenges
- Concluding remarks
- Data availability
- Competing interests
- Disclaimer
- Acknowledgements
- Financial support
- Review statement
- References




